14 Chapter 14: Industrial Enzymes and Their Applications in Food, Biofuel and Pharmaceutical Industries
Maedeh Mohammadi and Venkatesh Balan
Chapter Outline
14.2. Enzyme Nomenclature and Classification
14.3. Enzyme Activity and Function
14.4. Sources of Industrial Enzymes
14.5. Extraction and Purification of Enzymes
14.7. Screening for Novel Enzymes
14.8. Enzymes in the Food Industry
14.9. Enzymes in the Biofuel Industry
14.10. Enzymes in the Pharmaceutical Industry
Learning Outcomes
By the end of this chapter, you will be able to:
- Explain the fundamental properties of enzymes and their classification system
- Describe enzyme production, purification, and immobilization techniques
- Identify key enzyme applications in food processing and quality improvement
- Compare enzymatic processes in first, second, and third-generation biofuel production
- Evaluate the role of enzymes in pharmaceutical manufacturing and therapeutic applications
- Analyze the advantages of enzymatic approaches over traditional chemical processes
14.1. Introduction
Enzymes are protein catalysts that accelerate biological reactions with high specificity, interacting only with substrates that match the shape and chemistry of their active site. They are derived from animals, plants, and microorganisms and have an indispensable role in catalyzing biochemical reactions. Enzymes provide superior specificity, efficiency, and eco-friendliness compared to chemical reactions, reducing by-products and energy consumption. They are reusable as they remain unchanged during reactions. Enzymes are highly specific due to their active site and binding site. The active site binds the substrate and facilitates its chemical transformation through specific amino acid interactions, ensuring precise catalysis. The binding site, adjacent to or overlapping the active site, orients the substrate using non-covalent interactions like hydrogen bonds, opposite charge attractions, and van der Waals forces, stabilizing the enzyme-substrate complex for efficient catalysis.
In an industrial context, industrial enzymes refer to enzymes used extensively in various industrial processes to catalyze certain biochemical reactions. Today, enzymes are used to make more than 700 commercial products for the food and beverage, detergent, textile, biofuel and pharmaceutical industries (1). Enzymes have an extensive array of uses and are vital for enhancing the productivity and eco-friendliness of various manufacturing practices. These specialty enzymes can function under stringent industrial conditions such as temperature, pH, solvents, etc. In addition to their adaptability, these enzymes are characterized by high productivity and stability. Enzymes catalyze diverse reactions, including synthesis, cleavage, and conversion of compounds, by employing mechanisms such as acid-base catalysis, covalent bonding, or stabilization of transition states. This chapter provides a multifaceted overview of industrial enzyme technology and explores the various production methods, applications, advances in using genetic engineering tools, and transformative impact that these biocatalysts are exerting in key industries.
14.2. Enzymes Nomenclature and Classification
Originally, enzymes were given names that were not descriptive, such as:
- Rennin, which triggers the coagulation of milk during cheese production.
- Pepsin, which catalyzes protein hydrolysis under acidic pH conditions.
- Trypsin, which is known for protein hydrolysis under slightly alkaline pH conditions.
Subsequently, the method of naming enzymes evolved so that the suffix "-ase" was added either to the substrate compound with which the enzyme reacts or to the exact biochemical reaction that the enzyme catalyzes (2). For example:
Substrate name + ase
- Lactase is an enzyme that catalyzes lactose → glucose + galactose
- Lipase is an enzyme that catalyzes fat → fatty acids + glycerol
- Maltase is an enzyme that catalyzes maltose →glucose
- Urease is an enzyme that catalyzes urea + H2O →2NH3 + CO2
- α-Amylase is an enzyme that catalyzes starch → glucose + maltose + oligosaccharides Catalyzed reaction (second word in the name) + ase
- Glucose isomerase is the enzyme that catalyzes glucose → fructose
- Alcohol dehydrogenase is the enzyme that catalyzes ethanol + NAD+ → acetaldehyde + NADH
- Glucose oxidase is the enzyme that catalyzes D-glucose +O2 + H2O → gluconic acid
With the discovery of additional enzymes, the existing nomenclature system led to confusion, so the International Enzyme Commission drew up a clearer scheme in 1964. The revised enzyme nomenclature system groups enzymes into six principal classifications corresponding to the broad category of chemical reaction facilitated. Each major class also includes subclasses, sub-subclasses, and sub-sub-subclasses for even finer categorization. Accordingly, each enzyme is described by a sequence of four numbers preceded by EC (Enzyme Commission).
An enzyme database is a specialized repository that stores and organizes information about enzymes, their structures, functions, catalytic activities, and biochemical properties. These databases are essential tools for researchers in biochemistry, biotechnology, and molecular biology, as they provide comprehensive data on enzyme classification, reaction mechanisms, cofactors, substrates, and potential applications. The first digit represents the class, where six classes of enzymes are categorized based on the nature of the reaction. The second digit signifies the subclass, categorizing enzyme classes based on the groups or chemical bonds targeted by the enzyme. The third digit denotes the sub-subclass, indicating the coenzyme or cofactor necessary for enzyme activity. The fourth digit specifies the serial number of the individual enzyme within the list. As an example, the EC number of oxalate oxidase is shown and described in Fig. 14.1.

Figure 14. 1. Nomenclature of enzymes. Mohammadi and Balan (CC0 1.0).
Classification of Enzymes
Based on the type of reactions that enzymes catalyze, six categories (EC number classification) are considered for enzymes as follows:
- EC 1: Oxidoreductases: enzymes involved in oxidation-reduction reactions.
- EC 2: Transferases: enzymes involved in transferring functional groups from one molecule to another.
- EC 3: Hydrolases: enzymes involved in hydrolytic cleavage of C-O, C-C, C-N and some other bonds.
- EC 4: Lyases: enzymes involved in cleavage of C-O, C-C, C-N and other bonds by forming double bonds or rings other than hydrolysis and oxidation.
- EC 5: Isomerases: enzymes involved in all kinds of isomerization reactions
- EC 6: Ligases: Enzymes involved in joining two molecules with hydrolysis of ATP
Six classes of enzymes and their functions are summarized in Table 14.1.
Table 14.1. Classification of enzymes and the type of catalyzed reaction.
| Enzymes | Type and scheme of reaction catalyzed | Example |
|---|---|---|
| Oxidoreductases | Transfer of electron from one molecule to another | Dehydrogenases, Reductases, Oxidases, |
| Ared + Box⇌ Aox + Bred | Peroxidases | |
| Transferases | Transfer functional groups from one molecule to another | Amino transferase or Transaminase, |
| A-X + B ⇌ A + B-X | Kinase | |
| Hydrolases | Use water to cleave a bond | All digestive enzymes |
| A-B + H₂O ➞ A-OH + B-H | like α-amylase, and Lipase | |
| Isomerases | Control the conversion of one isomer to another by transferring groups within molecules | Phosphoglucomutase, Triphosphate isomerase, Phosphohexose isomerase |
| ABC ⇌ CAB | ||
| Lyases | Cleave chemical bonds without water | Aldolase, Fumarase, Argininosuccinase |
| X-A-B-Y ⇌ A=B + X-Y | ||
| Ligase | Formation of new bonds using ATP as an energy source | Glutamine synthetase, Pyruvate carboxylase, DNA ligase |
| A + B + ATP ➞ A B+ ADP + Pi |
Regardless of this classification of enzymes, enzymes could be involved in catabolic or anabolic reactions (Fig. 14.2).
- Catabolic reactions involve the decomposition of large molecular compounds into smaller components with the simultaneous release of energy. Examples of catabolic processes include digestion, in which complex nutrient molecules are broken down, and oxidative respiration, in which sugars are broken down to produce energy in the form of ATP. Such reactions are exergonic in nature but usually do not have the necessary activation energy to proceed without assistance. However, the presence of enzymes lowers the activation energy needed, so degradation reactions that otherwise have too little energy to start spontaneously in biological systems can proceed (3).
- Anabolic reaction is the process of building up large organic molecules from small molecules with some energy input. Synthesis of polysaccharides from simple sugars and proteins from amino acids are examples of anabolic reactions. Anabolic reactions are endergonic that do not occur spontaneously, so they require the extra energy input.
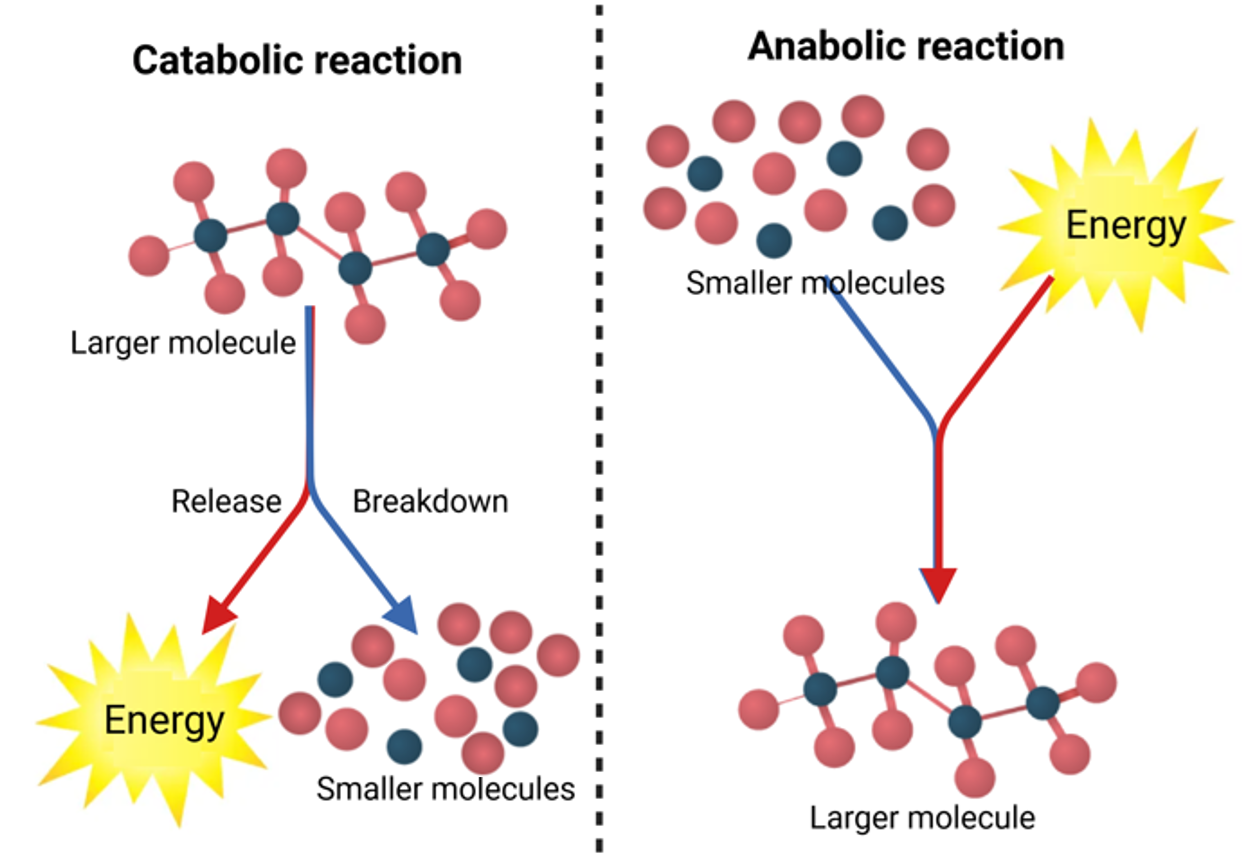
Figure 14.2. Schematic representation of catabolic and anabolic reactions. Mohammadi and Balan (CC0 1.0).
14.3. Enzyme Activity and Function
The most important property of enzymes is their ability to increase the rate of reactions, a property known as catalytic activity. When quantifying enzymatic reaction rates, typically either substrate depletion or product generation gets tracked over a defined timeframe. As per the International Union of Biochemistry, one international unit (IU) numerically represents enough enzyme to transform 1 μmol of substrate per minute. For instance, for cellulase activity, IU denotes the amount of enzyme that will liberate 1 μmol of glucose each minute, considering the polymeric cellulose substrate has variable molecular weight and the molecular weight of substrate cannot be defined precisely (2).
In general, spectrophotometric, fluorometric or chromatographic methods can be used to quantify the reactants/products and calculate the enzyme units. For example, in a spectrophotometric assay, the effect of the enzyme on the absorption of light at a specific wavelength can be measured (Fig. 14.3). When the enzyme catalyzes the conversion of the substrate into a product, the absorbance changes, allowing the reaction rate to be calculated. By comparing the initial and final concentrations of substrates or products, the amount of change per unit time can be determined and expressed as a reaction rate. This rate is a quantitative measure of enzyme activity and is fundamental to understanding enzyme kinetics, optimizing reaction conditions and evaluating the effects of various factors on enzymatic performance. The turnover number serves as a measure for comparing the catalytic capabilities of different enzymes. It indicates how many reaction molecules are converted into products by a single enzyme component within a certain period. It provides indirect information on the catalytic potency of an enzyme. Higher conversion values indicate a higher efficiency of the enzyme’s acceleration capacity.
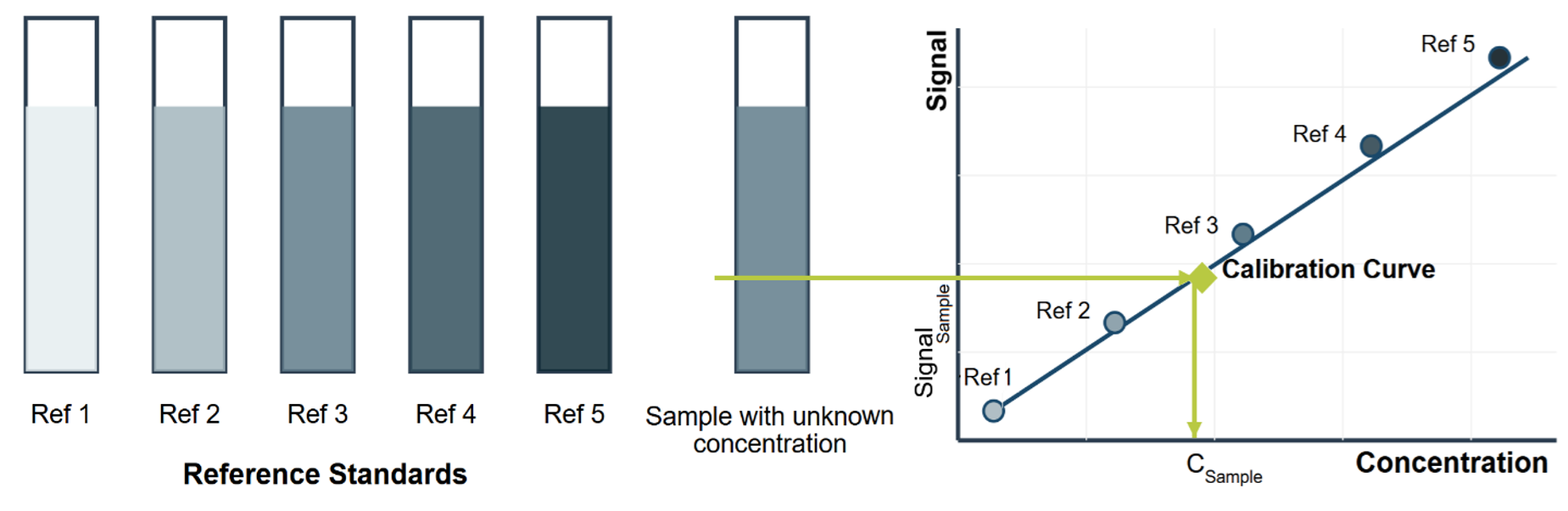
Figure 14.3. Spectrophotometric method for enzyme activity measurement showing reference standards with increasing concentrations (left) and the corresponding calibration curve used to determine the concentration of an unknown sample (right). Mohammadi and Balan (CC0 1.0).
Enzyme Active Site
An enzyme's active site is a specific region on the enzyme's surface where the substrate (the molecule being acted upon) fits, like a key in a lock. This site is typically a pocket or groove formed by particular amino acids arranged precisely. These amino acids work together to hold the substrate in place and accelerate the chemical reaction. The side chains of these amino acids extend into the pocket, enabling them to bind to and interact with the substrate at the correct locations. In this way, the enzymatic architecture provides a tailored scaffold to simultaneously recognize, stabilize and modify cognate bonds at this reactive hub. Each amino acid residue can be large or small, hydrophilic or hydrophobic, weakly basic or acidic, and negatively charged, positively charged, or neutral. These amino acid residues can act as acids or bases, facilitate the cleavage of substrates, or stabilize reaction intermediates.
The active site is highly selective for a specific substrate or a group of structurally related substrates. This specificity is crucial for the enzyme's role in catalyzing certain reactions. The unique structural features of the active site contribute to the specificity of the enzyme and ensure that it selectively interacts with its assigned substrate and promotes specific biochemical reactions. An originally postulated model to explain enzyme-substrate associations was based on a “lock-and-key” principle, if the morphological and topological features between the active site of an enzyme and the optimally paired substrate complement each other - analogous to a key fitting exactly into its lock. This theory emphasizes the synergy of molecular surface shapes that enable recognition. An alternative paradigm of “induced fit” states that enzymes can undergo slight dynamic conformational changes upon binding a substrate to improve the interface. The resulting transient enzyme-substrate complex represents a temporary energetic transition state that eventually relaxes into converted products and a restored enzyme. With each repeated catalytic cycle, the released enzyme regenerates its initial configuration to sequentially bind new substrates until the reactants are exhausted (Fig. 14.4). Accordingly, the enzyme action can be summarized in two steps:
- Step 1: Combination of enzyme (E) and the substrate (S). E+S → [ES]
- Step 2: Disintegration of the complex molecule to give the product (P). [ES]→ E+P
Thus, the overall catalytic action of enzymes: E + S → [ES] → [EP] → E + P
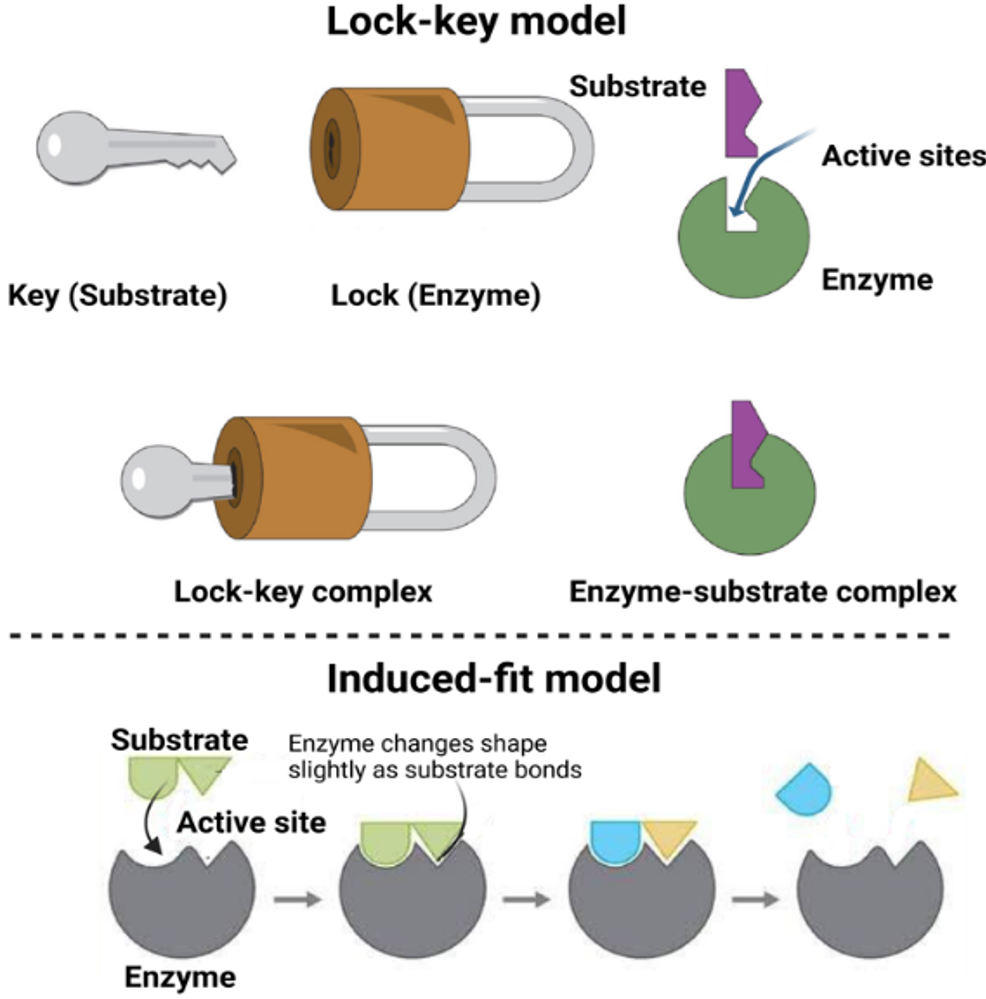
Figure 14.4. Enzyme-substrate binding models. Reprinted from Raval and Ganatra, (2022) (4), under CC BY-NC-SA 4.0 license.
Enzyme Specificity
Enzymatic catalysts have a strict specificity - they only accelerate certain types of reactions. The complex molecular topography of the activation pocket determines selectivity by sterically and chemically complementing compatible substrates and eliminating incompatible substrates that do not fit and align properly. The attraction between oppositely charged residues and reactants, coordinated hydrophobic and hydrophilic interactions, shape recognition and polarity matching enable tight binding orientation to activate the transition state for catalyzed structural transformations. As a result, enzymes not only stabilize reaction intermediates but also accelerate the achievement of equilibrium, where the conversion of substrates to products is balanced by the reverse reaction. Through numerous non-covalent affinity interactions, enzymes simply lower the kinetic barrier to allow rapid conversion in both directions.
Cofactors for Enzymes
Many enzymes require a non-protein component, a so-called cofactor, to assist them in their reaction. In this case, the inactive enzyme, the apoenzyme, must combine with the cofactor to form the active enzyme; this combination of an inactive apoenzyme with its non-protein component, which makes it active is called a holoenzyme (Fig. 14.5a).
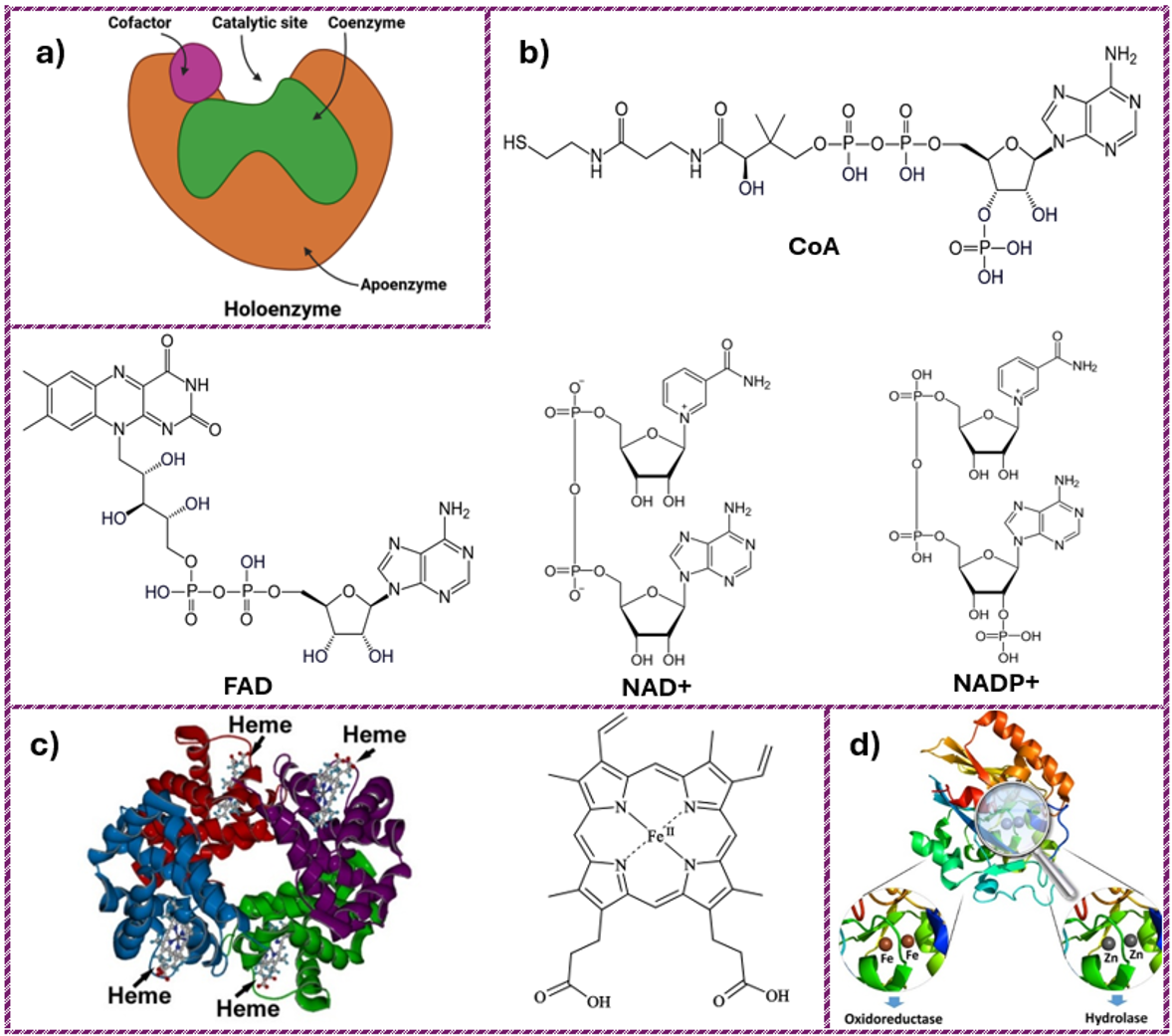
Figure 14.5. Enzyme structure and cofactors. Here, (a) Schematic of a holoenzyme showing the apoenzyme with catalytic site, coenzyme, and cofactor; (b) Chemical structures of common coenzymes including CoA, FAD, NAD+, and NADP+; (c) Quaternary structure of a heme-containing enzyme and heme chemical structure and (d) Inorganic metals as enzyme cofactors shown in metalloenzyme structures. Images (b) and (c) from Wikimedia Commons, licensed under CC BY-SA 2.0. Reproduced from Valasatava et al. (2018) (5), under the Creative Commons Attribution License (CC BY).
Cofactors prove to be indispensable for the functionality of enzymes, either as permanent innate components or as transient auxiliaries associated with the protein scaffold. Indispensable inorganic ions such as zinc, magnesium and iron often occupy specific structural sites and thus contribute to catalytic competence. Through this enzyme-cofactor synergy, chemical adjuvants extend the versatility and efficiency of enzymatic systems beyond what their native protein structures alone can accomplish. Cofactors can be categorized into three groups:
- Coenzymes: these are organic molecules such as Nicotinamide Adenine Dinucleotide (NAD+), Nicotinamide Adenine Dinucleotide Phosphate (NADP+), Flavin Adenine Dinucleotide (FAD), and Coenzyme A (CoA) that serve as carriers for chemical groups or electrons (Fig. 14.5b). They do not remain bound to the enzymes but only bind to an enzyme during catalysis and are then detached again. Most coenzymes are the derivatives of vitamins.
- Prosthetic groups: These are organic molecules that are always tightly bound to an enzyme to support the catalytic functions of enzymes. For example, heme is an iron-containing prosthetic group with an iron atom in the center and FAD is another prosthetic group present in many enzymes (Fig. 14.5c).
- Inorganic ions: For the catalysis of certain enzymes, an ion is required at the active site to form coordinative bonds. Ions such as Ca++, Mg++ and K+ etc. help to improve the speed of enzymatic reactions. For example, a chloride ion increases the salivary activity of amylase and zinc is a metal ion cofactor used by a few enzymes (Fig. 14.5d).
Effect of Environment on Enzyme Activity
Since most enzymes are proteins, their activity is influenced by various physical and chemical factors that either disrupt the protein structure or affect the reaction rate in general. These factors include the concentrations of the various components, including substrate, product, enzyme, cofactor, etc., and the reaction conditions, including pH, temperature and shear. These factors are discussed below.
Effect of substrate concentration
When a specific quantity of enzymes is available, the rate of an enzymatic reaction increases with increasing substrate concentration. Nonetheless, when the substrate concentration becomes exceedingly high, the enzymes reach a saturation level at which further increases in substrate concentration no longer increase the reaction rate. At this saturation threshold, the substrate abundance is such that almost all active sites of the enzyme are occupied by the substrate, leading to substrate saturation within the enzyme molecules. The excess substrate molecules can only participate in the reaction when the substrate already bound to the enzymes has undergone a reaction and product has been released, or when it is released without reaction. If the number of substrate molecules is greater than that of the free enzyme, the reaction rate remains the same and a plateau is observed in the reaction rate profile, as shown in Fig. 14.6a (6).
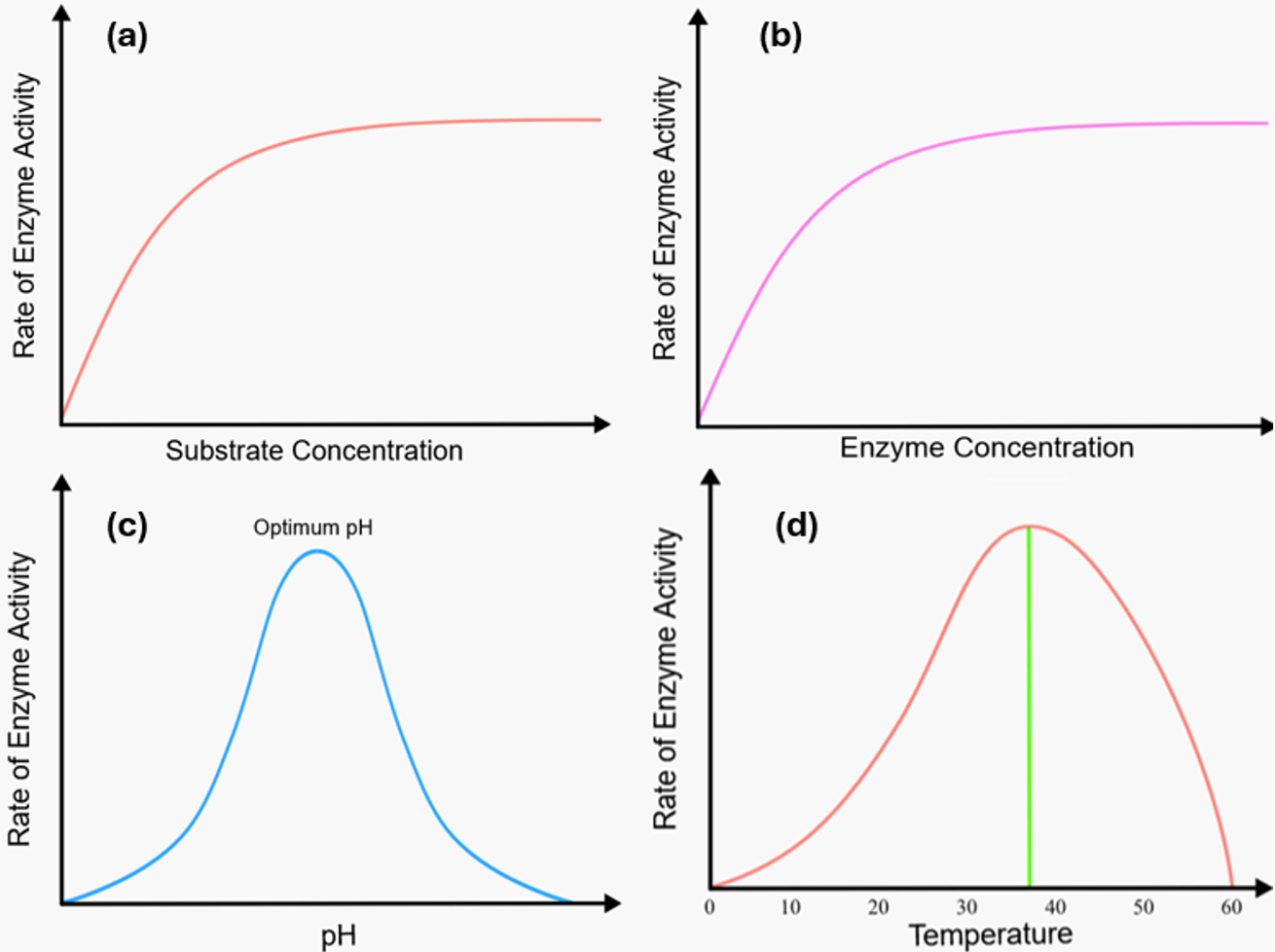
Figure 14.6. Factors affecting enzyme stability. Here, (a) Effect of substrate concentration on the reaction rate at a fixed enzyme concentration, (b) Effect of enzyme concentration on the reaction rate at a fixed substrate concentration, (C) Effect of pH on the rate of a reaction catalyzed by a fixed amount of enzyme, and (d) Effect of temperature on the rate of a reaction catalyzed by a fixed amount of enzyme. Mohammadi and Balan (CC0 1.0).
Effect of enzyme concentration
The effect of enzyme concentration on the reaction rate is like that of substrate concentration (Fig. 14.6b). If the enzyme concentration is significantly lower than the substrate concentration, the rate of the enzyme-catalyzed reaction remains sluggish. However, as the enzyme concentration increases, the reaction rate usually increases in parallel as additional active sites become accessible for substrate binding and facilitate the formation of more enzyme-substrate complexes. This phenomenon applies to all catalysts, as the reaction rate inevitably increases with increasing catalyst concentration. However, if all available substrate already has an enzyme to which they can bind, further increasing the enzyme concentration will not affect the reaction rate. At this point, additional enzymes no longer contribute, and the reaction rate reaches the maximum rate and the reaction rate profile plateaus. In industrial processes, determining the appropriate enzyme concentration is of fundamental importance. This helps to maximize the efficiency of enzyme-catalyzed reactions and minimize production costs.
Effect of pH
The pH of the reaction solution is one of the most influential parameters on enzyme activity, and enzymes exhibit optimum activity within a certain pH range. Extreme pH, whether high or low, has the potential to denature enzymes, resulting in a loss of its three-dimensional structure and thus its catalytic function, which is mostly irreversible. Since enzymes are proteins consisting of amino acid residues, they react sensitively to fluctuations in the hydrogen ion concentration or pH value. Even a slight change in pH can disturb the ionization status of the acidic and basic amino acids in the enzyme and substrate. It is crucial that the ionizable side groups in the active site retain specific charges to ensure effective binding between the enzyme and its substrate. Even neutralizing a single charge within these groups can lead to a significant change in the enzyme's catalytic activity. For example, glutamic acid is an acidic amino acid in the lower pH range with pK = 4.5, while lysine is a basic amino acid in the higher pH range with pK=10.0. Enzymatic activity depends on the specific charges carried by the amino acid residues in the active site. Consequently, the catalytic efficiency of the enzyme is directly influenced by the prevailing pH value. Consider, for example, an active center of an enzyme that contains a residue of each of the amino acid’s glutamic acid and lysine. For the enzyme to function optimally, both amino acids must maintain their charged state. Glutamic acid is charged at a pH value of 4.5, while lysine achieves this at a pH value of 10.0. Consequently, the enzyme shows its maximum activity within the pH range of 4.5 to 10.0. Enzymes usually show their maximum activity within a specific and relatively narrow pH range in which the molecules are present in their respective charged form. This relationship between the reaction rate and the pH value is represented by a bell-shaped curve, with the central point being referred to as the optimum pH value of the enzyme (Fig.14.6c).
Most body fluids, except for gastric juice, maintain a pH range between 6 and 8, which corresponds to the optimal activity range of most enzymes. For example, salivary amylase shows optimal activity at a pH of 6.8, while pancreatic chymotrypsin functions optimally in the slightly alkaline range of pH 7 to 8. However, some enzymes have optimal pH values that lie outside this typical range. Pepsin, for example, an enzyme active in the stomach, shows its highest activity at a pH value between 2.0 and 3.3 (2). Understanding the pH dependence of enzymes is crucial for optimizing their performance in various applications, e.g., in industrial processes or laboratory experiments. Adjusting the pH within the optimal range ensures the highest enzyme activity and consequently the most efficient catalysis of biochemical reactions.
Effect of temperature
According to the Arrhenius equation, the rate of a chemical reaction depends on the temperature:
k= A0e-E/RT
So, if lnk is plotted versus 1/T, the slope will give E/R from which the activation energy is determined. The Arrhenius equation effectively characterizes the temperature sensitivity of numerous reactions catalyzed by enzymes. With increasing temperature, the reaction rate increases, driven by the higher energy levels and the greater mobility of atoms within the enzyme molecules. A widely accepted guideline in chemistry is that an increase in temperature of about 10°C usually leads to a doubling of the reaction rate in most chemical reactions. This basic concept also applies to enzymatic reactions. However, above a certain threshold, increasing the temperature can lead to a decrease in the reaction rate (Fig. 14.6d). This phenomenon occurs when the protein structure is denatured, and the active site is disturbed due to the increased motion of the amino acid residues, which starts to tear apart the protein structure. Denaturation results from the unfolding of the protein chain due to the disruption of weak bonds, such as hydrogen bonds, which leads to a reduction in the overall reaction rate. Denaturation generally occurs in the temperature range of 45°C to 55°C for many proteins. Although it may appear that an enzyme has optimal reaction rates between 40°C and 50°C, practical biochemical reactions often take place at lower temperatures. This is because enzymes that are unstable at higher temperatures undergo denaturation after a short time, usually within a few minutes. Yet, some enzymes, especially those derived from thermophilic organisms that thrive in particularly hot environments, exhibit considerable resistance to denaturation induced by high temperatures.
Enzyme-catalyzed reactions approach nearly zero rates at extreme temperatures of 0°C and 100°C, leading to several practical applications. Sterilization uses boiling water to effectively denature the enzymes of bacteria on and in objects. When preserving food, refrigeration or freezing techniques are used to slow down enzyme activity. During winter, the body temperatures of hibernating animals drop, reducing metabolic processes to a level that can be maintained by the energy stored in fat reserves.
Effect of shear stress
Shear stress, which refers to the force per unit area acting parallel to the surface of a material, is thought to impair enzyme activity. Shear stress can cause a disruption in the intricate structure of the enzyme molecule, leading to changes in the tertiary or quaternary structure of the enzyme. High shear stress can potentially cause denaturation or unfolding of the enzyme, ultimately leading to denaturation. This is particularly important for shear-sensitive enzymes. The typical mechanical force acting on an enzyme solution, especially in bioprocessing or other industrial applications, is liquid shear, which is generated by processes such as flowing a liquid, stirring with an agitator or shaking a vessel.
Understanding the effect of shear on enzyme stability is important for enzyme reactor design, especially considering that the contents of the reactor must be stirred or shaken to reduce mass transfer resistance. Many industrial enzymes, such as those used in fermentation or biofuel production, may be subject to shear stress due to mechanical shaking, stirring or pumping of liquids. Therefore, bioprocess developers and enzyme engineers should consider shear sensitivity in process design. Strategies include the selection of shear-resistant enzymes or the optimization of process conditions to minimize shear effects. Immobilizing enzymes on solid supports or encapsulating them in protective matrices can also be used to protect them from the deleterious effects of shear stress. When designing bioreactors, many considerations, such as the design of the agitator, agitation speed, etc., should be considered to avoid excessive shear stress on the enzymes. Accordingly, efficient mixing while minimizing shear forces to maintain optimal enzyme activity and a balance between these two factors is very important in various industrial applications.
Since enzymes are proteins, whose activity relies on the precise sequence of amino acids and the intricate tertiary structure of the hundreds of amino acids that make up the protein, it is not feasible to chemically manufacture enzymes on a large scale. Typically, enzymes are derived from microorganisms cultivated in pure culture or directly extracted from plants and animals (Fig. 14.7). Microorganisms, including bacteria and fungi, are the most important sources of industrial enzymes. Certain species of bacteria, including Bacillus, are prolific producers of enzymes such as amylases, proteases and lipases. Filamentous fungi such as Trichoderma and Aspergillus are used extensively to produce enzymes such as cellulases, amylases and xylanases. Enzymes can also be extracted from plants, such as bromelain and papain from pineapple and papaya, respectively, which are used in food processing and pharmaceuticals. Another example of a plant-based enzyme is lipase extracted from the seeds of certain oil-producing plants such as castor beans. While plant-based enzymes are less common for industrial applications compared to enzymes derived from microorganisms, they may be preferred for certain applications due to their ability to make products that enzymes from animal or microbial sources either cannot make at all or cannot make under the conditions they encounter. Since plant-based digestive enzymes are effective in a broad pH range between 3.0 and 9.0, which is very well adapted to the human gastrointestinal environment, these enzymes are suitable for supporting broad digestive health applications (7). Animal enzymes, particularly those derived from the pancreas, such as trypsin and chymotrypsin, are used in the pharmaceutical and food industries. Animal rennet, a commercial extract containing the active enzyme rennin, also known as chymosin, is another example of animal-derived enzyme with extensive application in the dairy industry.
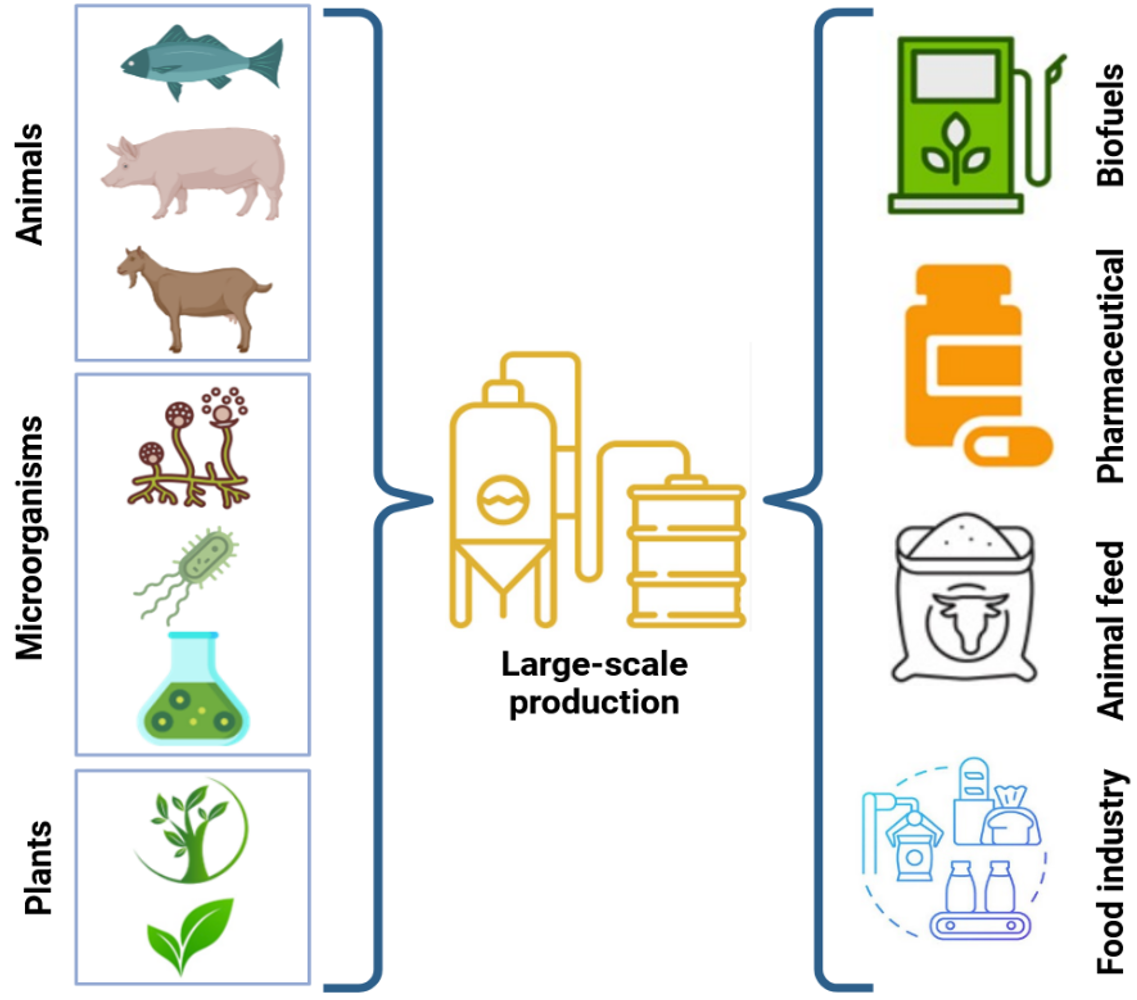
Figure 14.7. Sources of industrial enzymes (animals, microorganisms, plants) and their major applications in biofuels, pharmaceuticals, animal feed, and food industries. Mohammadi and Balan (CC0 1.0).
Microbes are the most important production platform for enzymes on an industrial scale, as they have a higher yield, stability and kinetics compared to plant and animal analogs. In addition, microbial enzyme systems can be easily adapted by genetic manipulation of simple fermentation hosts that exhibit great biochemical diversity. Such recombinant strains, developed since the advent of genetic engineering in the 1970s, now account for about 90% of commercial enzymes (8). They offer customized specificities, sensitivities and production quantities to meet cost targets. Looking to the future, new genome editing techniques promise further optimization of industrially relevant microbial enzyme factories.
Microbial Enzyme Production
In microbial enzyme production, microorganisms such as bacteria, fungi and yeasts are cultivated to produce enzymes on a large scale. Microbial enzyme production is a complex process in which various factors play a role, such as the selection of suitable microorganisms, optimization of culture conditions and subsequent processing. In general, two main approaches are used for microbial enzyme production: (1) submerged fermentation (SmF) and (2) solid-state fermentation (SSF), which are discussed here (Fig. 14.8).
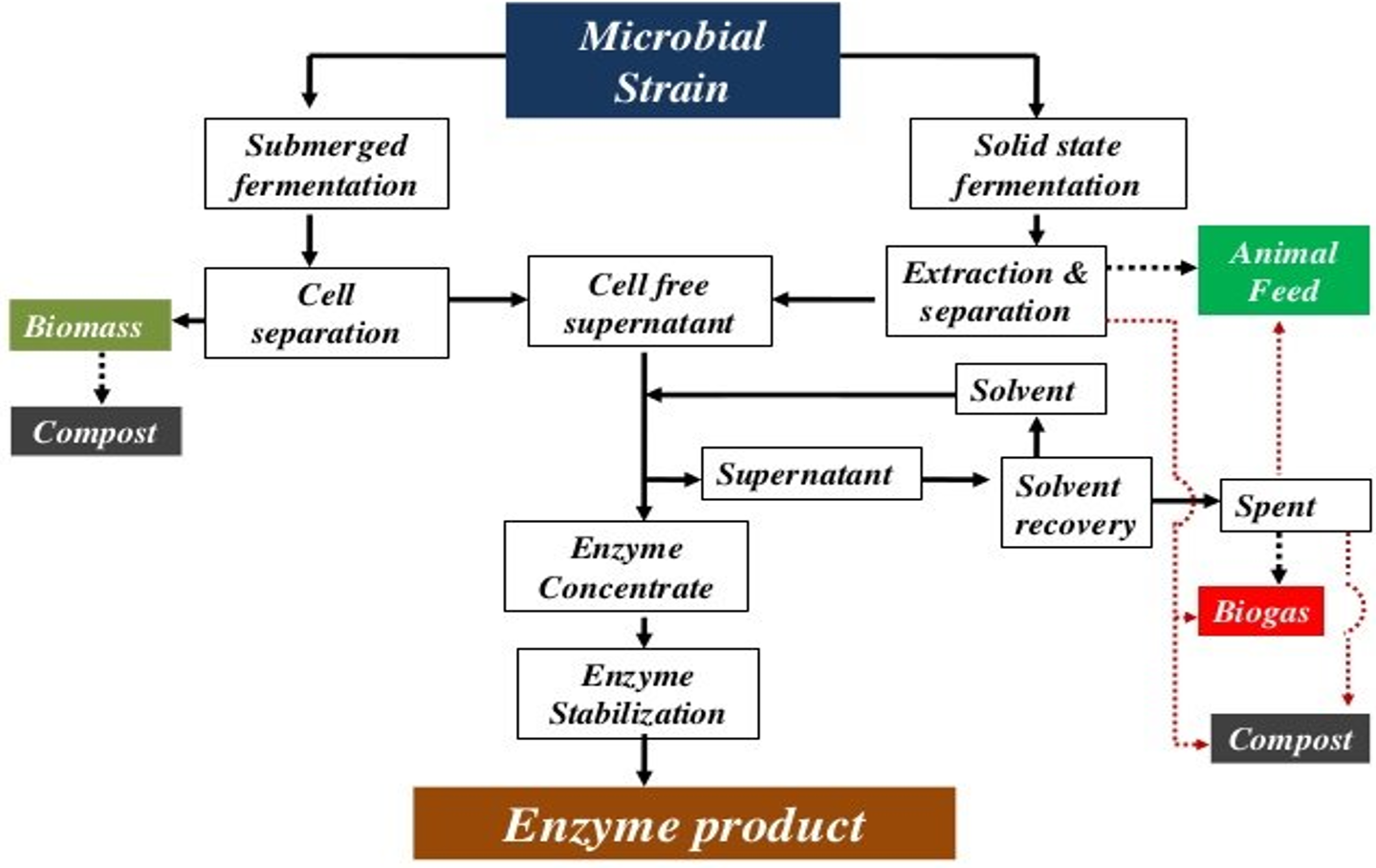
Figure 14.8. Process flow chart for enzyme production through solid-state fermentation and submerged fermentation. Adapted from ILRI (2011) (9), licensed under CC BY-NC-SA 3.0.
In SMF, microbes are propagated in a liquid medium to secrete enzymes directly into the surrounding broth. Such aqueous cultivation proves to be particularly suitable for bacteria and other microflora that require a lot of moisture for their growth and metabolism. Typical nutrient streams are soluble sugars, juices, molasses and formulated liquid media. The SmF infrastructure enables batch, semi-continuous or continuous operating modes. In fed-batch systems, sterile nutrients are fed intermittently to the active fermenter to maintain production. In continuous mode, fresh sterilized medium is fed to the culture while a portion of the culture is continuously withdrawn at the same flow rate at which it enters the fermenter.
SSF offers an alternative method for enzyme production in which microorganisms are cultivated on solid substrates without free water, with only a minimum amount of water available to maintain microbial growth and metabolic functions. While bacteria, yeasts and fungi can thrive on solid substrates, filamentous fungi exhibit exceptional compatibility with SSF. In general, agro-industrial residues such as wheat bran, rice bran or other lignocellulosic materials serve as suitable nutrient sources. SSF is usually operated in a batch process and offers the advantage of concentrated enzyme production.
Table 14.2 provides an overview of the comparison between SmF and SSF, including the various aspects of the two approaches and their advantages and disadvantages. Regardless of the fermentation conditions applied, SSF or SmF, the adjustment of various parameters such as pH, temperature, aeration and nutrient composition is necessary to increase the yield of enzyme production and improve the overall efficiency of microbial enzyme production (10).
Table 14.2 Comparison between solid-state and submerged fermentation (10).
| Aspect | Solid-state fermentation (SSF) | Submerged fermentation (SmF) |
|---|---|---|
| Definition | Process occurs in a solid matrix with minimal free water | Process utilizes liquid substrates with free-flowing water |
| Moisture requirement | Low moisture content is required for microbial activity and growth | High moisture content is required for optimal microbial activity and growth |
| Substrate form | Solid substrates, often agricultural wastes or lignocellulosic wastes | Liquid substrates, such as broths, molasses, fruit/vegetable juices, or wastewater |
| Enzyme release | Enzyme is produced and retained within the solid matrix | Enzyme is released directly into the fermentation broth |
| Suitable microorganism | Filamentous fungi, certain yeasts, and bacteria | Bacteria and microorganisms demanding high moisture content |
| Nutrient utilization | Nutrients are obtained from solid substrate through diffusion and enzymatic reaction | Nutrients are supplied in liquid form, readily available for microbial uptake |
| Fermentation mode | Mainly operated in batch mode | Operates in various modes: batch, fed-batch, continuous |
| Advantages | Minimal water and energy requirements, concentrated products | Enhanced mass transfer, better control of process parameters |
| Disadvantages | Limited scale-up potential, challenging control of process parameters | Higer water and energy requirements, potential contamination risks |
| Applications | Enzyme production, bioconversion of agricultural waste, bioremediation | Industrial enzyme production, pharmaceuticals, food processing, bioremediation |
14.5. Extraction and Purification of Enzymes
Extraction and separation processes for the purification of biological products, including enzymes, from microbial cultures are referred to as downstream processing. The extraction and purification of microbial enzymes involves several steps to obtain a pure and active enzyme preparation. When it comes to intracellular enzymes, the cells are obtained by centrifugation or filtration of the fermentation medium. These separated cells are subjected to an appropriate cell disruption method in a suitable buffer to release the intracellular enzymes. Several techniques exist for this purpose, including chemical methods such as detergents and chaotropic agents, physical methods such as homogenization and bead milling, and enzymatic methods such as treatment with lysozyme. On the other hand, if the enzyme is extracellular, the fermented medium itself serves as the enzyme source. To separate cell debris and insoluble particles from the crude extract, filtration and centrifugation are used to obtain a clear supernatant. The next step is enzyme separation by salt precipitation with ammonium sulfate or other salts, pH precipitation or precipitation with organic solvents to partially purify the enzyme by removing impurities.
Membrane-based separation techniques: Membrane filtration has revolutionized protein purification by providing efficient, scalable processes that maintain enzyme activity under mild processing conditions. Unlike conventional chromatographic methods, membrane-based separations operate primarily on size-based principles while allowing continuous processing with high throughput (11,12).
- Microfiltration (MF): it employs membranes with pore sizes ranging from 0.1-10 μm, making it ideal for initial clarification steps that separate cells and cell debris from soluble enzymes. For extracellular enzymes, MF effectively removes microbial cells from fermentation broths while allowing proteins to pass through the membrane. Various configurations include surface filters, depth filters, and rotary drum vacuum filters, each offering specific advantages depending on the particulate characteristics and viscosity of the process stream.
- Ultrafiltration (UF): it utilizes membranes with smaller pore sizes (1-100 nm) and is particularly valuable for concentrating enzyme solutions and buffer exchange. UF can achieve concentration factors of 10-100X while operating at low pressures (1-10 bar), minimizing the risk of enzyme denaturation. Advanced UF techniques include charged membranes that combine size exclusion with electrostatic interactions, significantly enhancing selectivity for enzymes with specific charge characteristics. Two-stage UF strategies using membranes with different molecular weight cut-offs (MWCOs) have proven effective for separating enzymes of different sizes, such as α-lactalbumin from β-lactoglobulin and various proteolytic enzymes.
- Dialysis: it represents another important membrane-based technique frequently used in enzyme purification. In this process, the enzyme solution is placed in a semi-permeable membrane bag or cassette with a specific molecular weight cut-off (MWCO) and immersed in a large volume of the desired buffer. Small molecules and ions diffuse across the membrane until the equilibrium is reached, while the larger enzyme molecules remain contained (Fig. 14.9). Dialysis is particularly valuable following salt precipitation steps, allowing gentle removal of ammonium sulfate or other salts that might interfere with subsequent purification steps or enzyme activity assays. While traditional dialysis can be time-consuming, modern cassette systems and continuous-flow dialysis have significantly improved processing times.
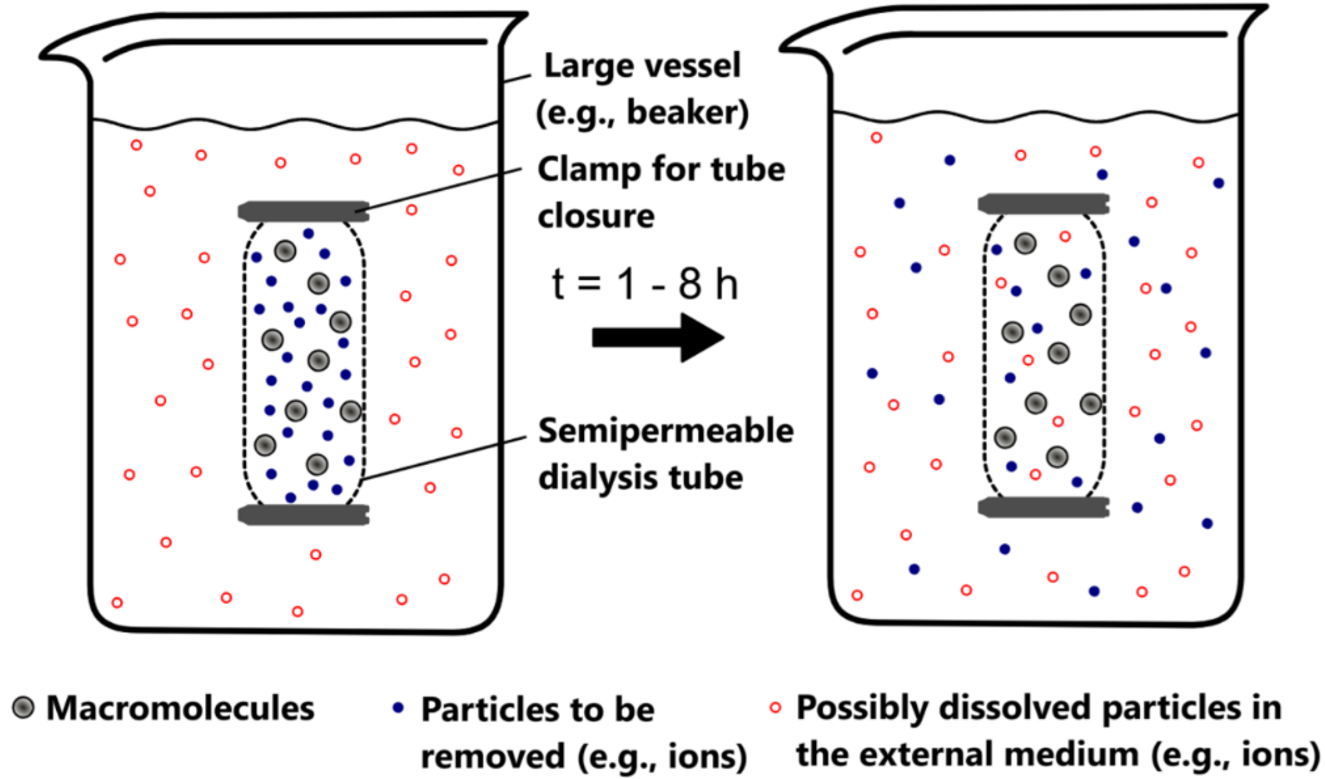
Figure 14.9. Principle of dialysis for enzyme purification. Left: Initial state with macromolecules (gray) and particles to be removed (blue) inside a semi-permeable dialysis tube suspended in a large vessel containing the outer medium with dissolved particles (red). Right: After 1-8 hours, small molecules diffuse across the membrane until equilibrium is reached, while larger macromolecules remain contained within the dialysis tube. Image from Wikimedia Commons by Andi schmitt, licensed under CC BY-SA 2.0.
- Tangential flow filtration (TFF): also known as crossflow filtration, has become particularly important for processing industrial enzymes. In TFF, the feed flows parallel to the membrane surface while filtration occurs perpendicular to the flow, significantly reducing membrane fouling (Fig.14.10). One drawback of TFF is its high initial flux, which can rapidly create a fouling layer if transmembrane pressure is not well-controlled. High-Performance Tangential Flow Filtration (HPTFF) exploits differences in both size and charge characteristics of proteins, enabling separation of enzymes that differ by less than three-fold in molecular weight through careful optimization of buffer conditions and membrane properties.
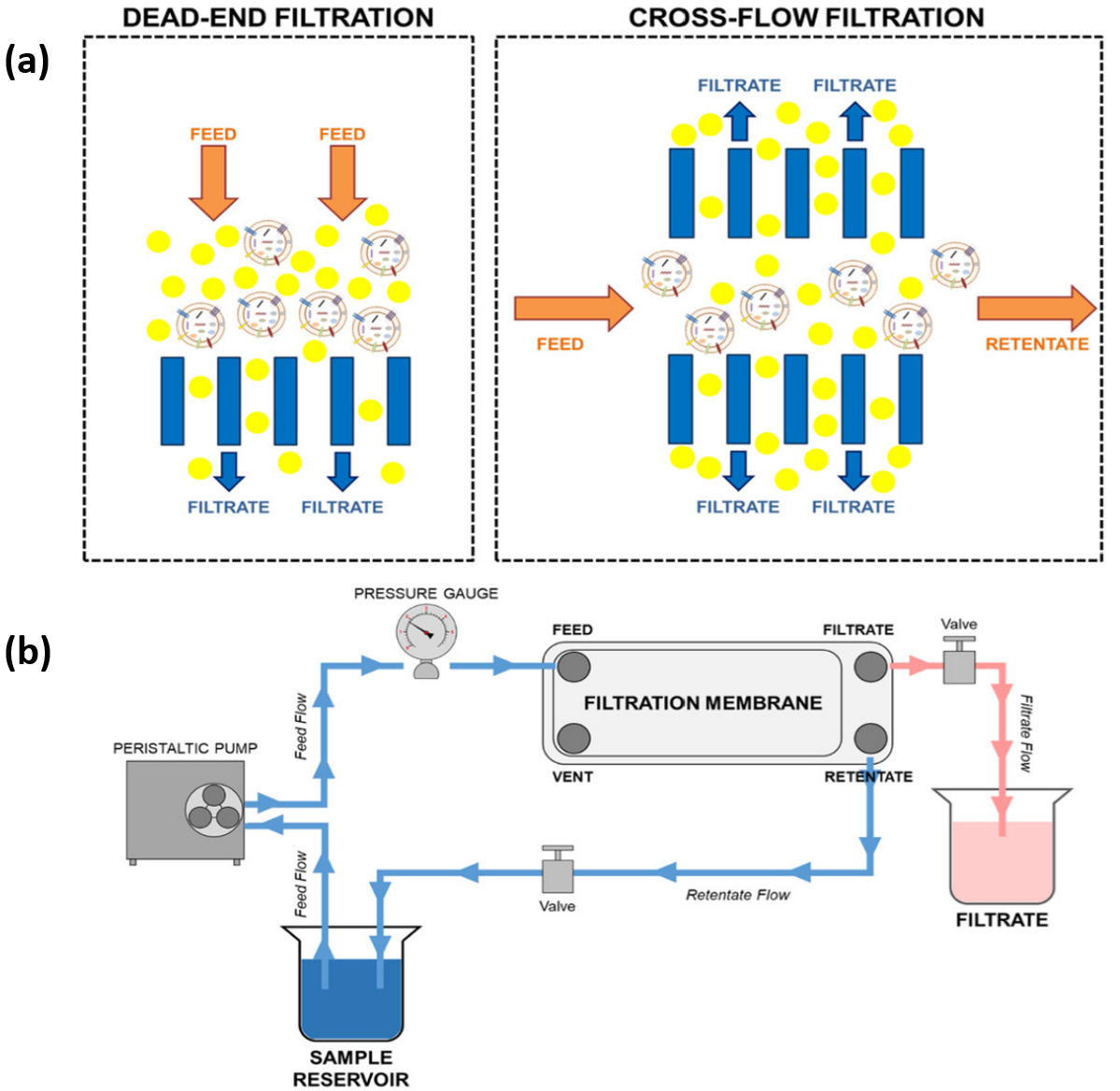
Figure 14.10. Comparison of filtration systems. (a) Dead-end versus crossflow filtration methods. (b) Schematic of tangential flow filtration system showing sample circulation through filtration membrane with separate filtrate and retentate flows. Image from Visan et al. (2022) (13), licensed under CC BY-NC-ND 4.0.
- Diafiltration: it is a variation of ultrafiltration, particularly useful for buffer exchange and desalting of enzyme preparations. This technique effectively removes low molecular weight impurities and salts following precipitation steps, providing a faster alternative to traditional dialysis. Diafiltration serves as an excellent alternative to gel filtration or dialysis for removing ammonium sulfate from protein preparations precipitated by the salt.
- Reverse osmosis (RO): it is an important membrane technique with pore sizes around 0.0001 μm, it is generally not applicable for enzyme purification due to the much larger size of protein molecules. RO is primarily used for water purification and concentration of small molecules, whereas enzymes typically range from 10 kDa to several hundred kDa in size, making them far too large for RO membranes. Instead, RO finds application in downstream processing for the concentration of small molecule products and water recovery from process streams.
Chromatographic Purification Methods
Depending on the properties of the enzyme and the desired degree of purity, a suitable chromatographic process is used to further purify the enzyme (Fig. 14.11). These purification methods include:
- Gel permeation chromatography: This method is also called size exclusion chromatography, works on the principle of size-specific separation. The solution containing the impure protein is introduced into a column filled with polymer beads. Larger protein molecules pass around the beads with ease and quickly leave the column. Smaller molecules and ions, on the other hand, must navigate through the polymer network within the beads, taking a longer route through the column.
- Ion exchange chromatography: Here separation of enzymes is based on their ionic interactions. The stationary phase consists of a resin with ionic functional groups that retain molecules with opposite charges. When the solution containing impure protein is introduced into a column filled with polymer beads carrying charged groups, proteins with an overall negative charge are attracted to resin beads carrying a positive charge, while non-interacting proteins are flushed. By changing the pH or salt concentration of the solution flowing through the column, the attracted proteins can then be eluted. This process can effectively separate different proteins in a mixture by adjusting the pH when loading the column and the pH or salt concentration when washing the column.
- Affinity chromatography. Enzymes are separated according to their binding affinity to a specific small molecule that is covalently bound to a stationary matrix. When the column is loaded with an impure protein solution, the proteins selectively bind to the molecules immobilized on the polymer, while the impurities are allowed to flow through. The pure protein is then extracted or eluted from the column.
- Hydrophobic interaction chromatography (HIC). In these methods proteins are separated according to their hydrophobic interactions. The stationary matrix contains hydrophobic ligands that initially interact with the hydrophobic regions of the protein molecules and cause them to localize to each other. The interaction between hydrophobic proteins and the resin is modulated by the composition of the running buffer. Higher salt concentrations increase this interaction, while lower salt concentrations decrease it, leading to the elution of the proteins from the matrix.
To assess the purity of the separated enzyme and to ensure that the enzyme meets the specifications required for industrial applications, electrophoresis is used next. The concentration of the purified enzyme is then increased by vacuum evaporation or precipitation to reduce the volume of the enzyme solution.
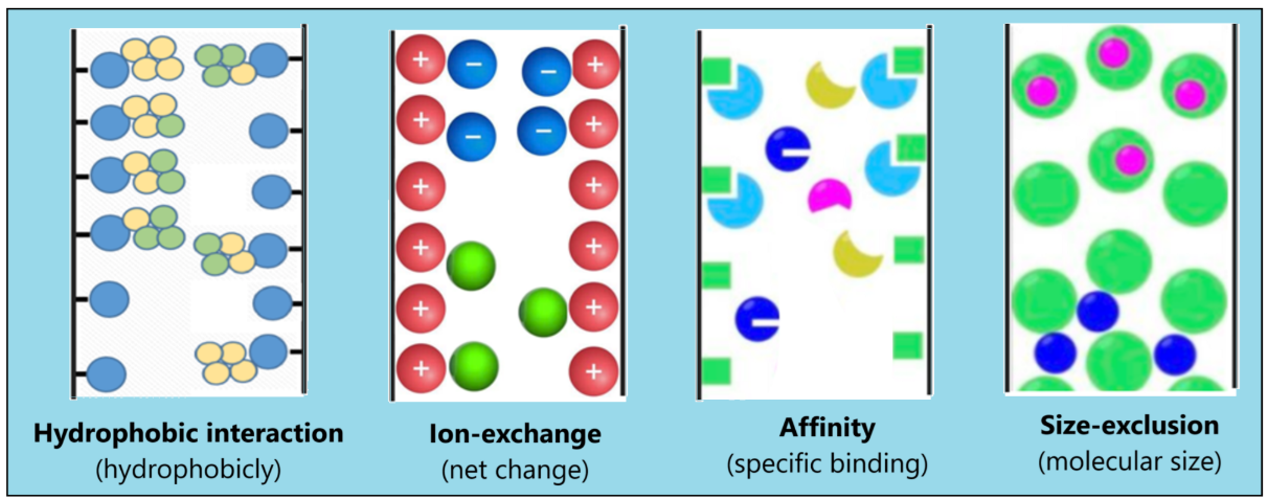
Figure 14.11. A scheme of enzyme purification techniques. Mohammadi and Balan (CC0 1.0).
For the next step in enzyme processing, which involves the evaluation of product purification and determination of biological activity, SDS-PAGE (sodium dodecyl sulfate polyacrylamide gel electrophoresis) can be used to confirm the absence of impurities. Methods such as the Bradford assay can be used to determine the protein concentration in the purified sample. To gain additional insight into the molecular properties of the enzyme, techniques such as mass spectrometry or western blotting can be used. To confirm that the enzyme retains its biological activity after the purification process, the specificity of the enzyme towards its substrates is evaluated to ensure that it behaves as expected. To increase the shelf life and stability of the enzyme product, preservatives such as sodium azide or thimerosal can be added to inhibit microbial growth and prevent contamination during storage. For enzymes that are susceptible to denaturation during freeze-thaw cycles, cryoprotectants such as glycerol or trehalose can be added prior to freezing to improve stability.
In the final stage, the enzyme is prepared in a form that improves its stability, ease of handling and applicability for specific industrial purposes. Common forms of the final enzyme product include liquid solutions (often stored in a buffered solution), spray-dried powders, pelletized products or encapsulated formulations (encapsulated in a protective matrix such as microspheres or microcapsules). The choice of the final enzyme form depends on factors such as the stability of the enzyme, the intended application and the requirements of the end users, and conditions must be carefully optimized to maintain enzyme activity and quality. Fig. 14.12 shows a general flow chart for microbial enzyme production and purification (3).
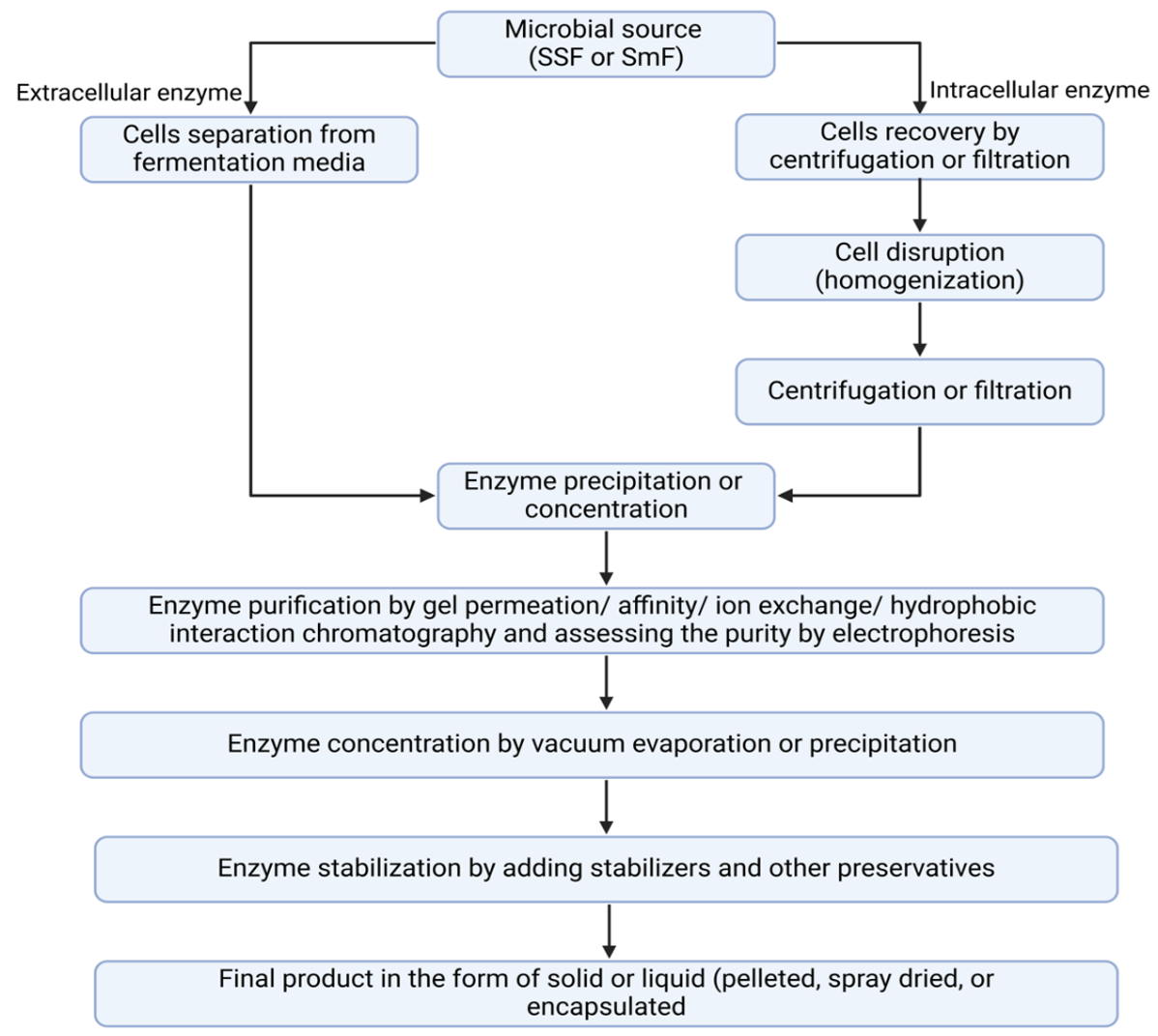
Figure 14.12. A general flow chart for enzyme purification from microbial cultures. Mohammadi and Balan (CC0 1.0).
14.6. Enzyme Immobilization
Since most enzymes are protein-based, they are soluble in water, which makes it difficult to separate and reuse in processes. To overcome this obstacle, enzymes can be immobilized in their soluble state either on the surface or in an insoluble matrix or in a semi-permeable membrane. Immobilized enzymes offer the decisive advantage that they can be reused, as they can be easily separated from the reaction solution and kept in a flow reactor. Furthermore, immobilized enzymes are more stable and easier to handle compared to their free counterparts. In addition, immobilized enzymes enable enzymatic reactions in non-aqueous environments, with solid support reinforcing the enzyme components, thereby increasing their catalytic activity and facilitating multiple reuses.
The main role of the carrier material is to reinforce the enzyme structures and thus maintain their functionality under unfavorable environmental conditions such as extreme pH or high temperatures. Various natural and synthetic carrier materials have been studied for their effectiveness in immobilizing enzymes and offer a range of options (14):
- Natural polymers: alginate, chitosan and chitin, collagen, carrageenan, gelatin, cellulose, pectin, starch, Sepharose, etc.
- Synthetic polymers: ion exchange resins/polymers, polyvinyl chloride, polyurethane microparticles, polyaniline, glutaraldehyde-activated nylon, UV-activated polyethylene glycol, etc.
- Inorganic materials: zeolites, ceramics, celite, silica, glass, activated carbon, charcoal.
Immobilization techniques can be divided into chemical and physical methods. Chemical immobilization is based on the formation of covalent bonds, while physical immobilization is based on the formation of non-covalent bonds.
Chemical immobilization techniques
- Covalent bonding: In the covalent binding method for enzyme immobilization, a stable covalent bond is established between the enzyme molecules (via a non-essential amino acid residue) and a support matrix (e.g. polyacrylamide, agarose, porous glass, porous silica, etc.). This technique is based on the creation of robust and durable bonds, usually through coupling agents or functional groups on the enzyme and the support material. The advantage of covalent binding is the improved stability and longevity of the immobilized enzymes, allowing them to be used over a longer period in various applications. This method also minimizes the leaching of the enzymes and ensures that the biocatalysts remain firmly attached to the carrier, resulting in increased reusability and efficiency. However, this method also has disadvantages, such as complicated procedures and long incubation times for proper immobilization, the need for structural modifications to covalently immobilize the enzyme by attaching it to a solid matrix, which can lead to denaturation of the enzymes and the small number of immobilized enzymes on the supports. Nevertheless, the advantages of this approach, including increased enzyme stability and efficient recovery/reuse, outweigh the disadvantages (15).
- Cross-linking: In cross-linking immobilization, covalent bonds are established between enzymes without the use of a carrier. In this approach, linkers are used as connectors between neighboring enzyme molecules, creating stable bonds and increasing overall stability. Bi-/multifunctional reagents such as glutaraldehyde, bisdiazobenzidine and hexamethylene diisocyanate are used as crosslinkers, with glutaraldehyde being the most used due to its bifunctional nature and low molecular weight. Various cross-linking methods have been used to date, including cross-linked spray-dried enzymes, cross-linked soluble enzymes and cross-linked aggregates (16). Cross-linking immobilization provides a robust connection between the enzymes, resulting in higher stability.
Physical immobilization techniques
- Adsorption: Adsorption is the simplest type of immobilization of enzymes in which reversible immobilization is achieved. Enzymes can be physically adsorbed onto a surface-active adsorbent by bringing an aqueous enzyme solution into contact with an adsorbent. Adsorption results from a weak binding force, such as hydrophobic interactions, hydrogen bonds, ionic bonds and salt bonds, between the carrier and the enzyme. Various adsorbents such as alumina, carbon, cellulose, ion exchange resins and others are commonly used for the immobilization of enzymes. However, not all types of enzymes are compatible with every carrier material. Successful adsorption requires the presence of specific active groups on the carrier material to facilitate interaction with the enzymes. In cases where such functional groups are lacking, carrier modifiers can be introduced to create links between the enzymes and carriers. Although adsorption techniques are simple, reversible and preserve the enzymes, the state of immobilization is very sensitive to factors such as temperature, pH of the solution and ionic strength. As a rule, the amount of enzyme loaded onto one unit of carrier is relatively small and the binding strength is rather weak.
- Entrapment: Entrapment is an irreversible method of immobilization in which the enzymes are entrapped in gels or fibers by covalent or non-covalent bonds. Cross-linked polymers, such as polyacrylamide gels, are commonly used for enzyme entrapment. In contrast to other methods, the enzymes do not undergo chemical reactions with the polymers during entrapment, minimizing the risk of denaturation. With this approach, the intrinsic properties of the enzyme are preserved, even though inactivation of the enzyme may occur during gel formation. Despite its advantages, enzyme entrapment also has its limitations. These include the limited extent to which enzymes can be entrapped, the potential destruction of the carrier during polymerization and the risk of enzyme leakage if the pore size of the carrier is too large. Techniques such as photopolymerization, electro polymerization and the sol-gel method are used to facilitate the entrapment of enzymes while overcoming these challenges (16).
- Encapsulation: The technique of interfacial polymerization enables the immobilization of enzymes in semipermeable membrane microcapsules. In this process, an organic solvent containing a copolymer component and a surfactant is stirred while an aqueous enzyme solution is introduced. A polymer membrane forms at the interface between the two liquids, effectively encapsulating the enzymes to protect them from the environment and maintain their activity over time. Despite its advantages, such as the large surface area, this technique is not without its limitations. Challenges such as diffusion issues and the potential rupture of the membrane due to rapid product accumulation need to be addressed. A scheme of chemical and physical immobilization techniques used for enzyme immobilization is given in Fig. 14.13 (15). Table 14.3 presents a summary of immobilized enzymes for food applications (16).
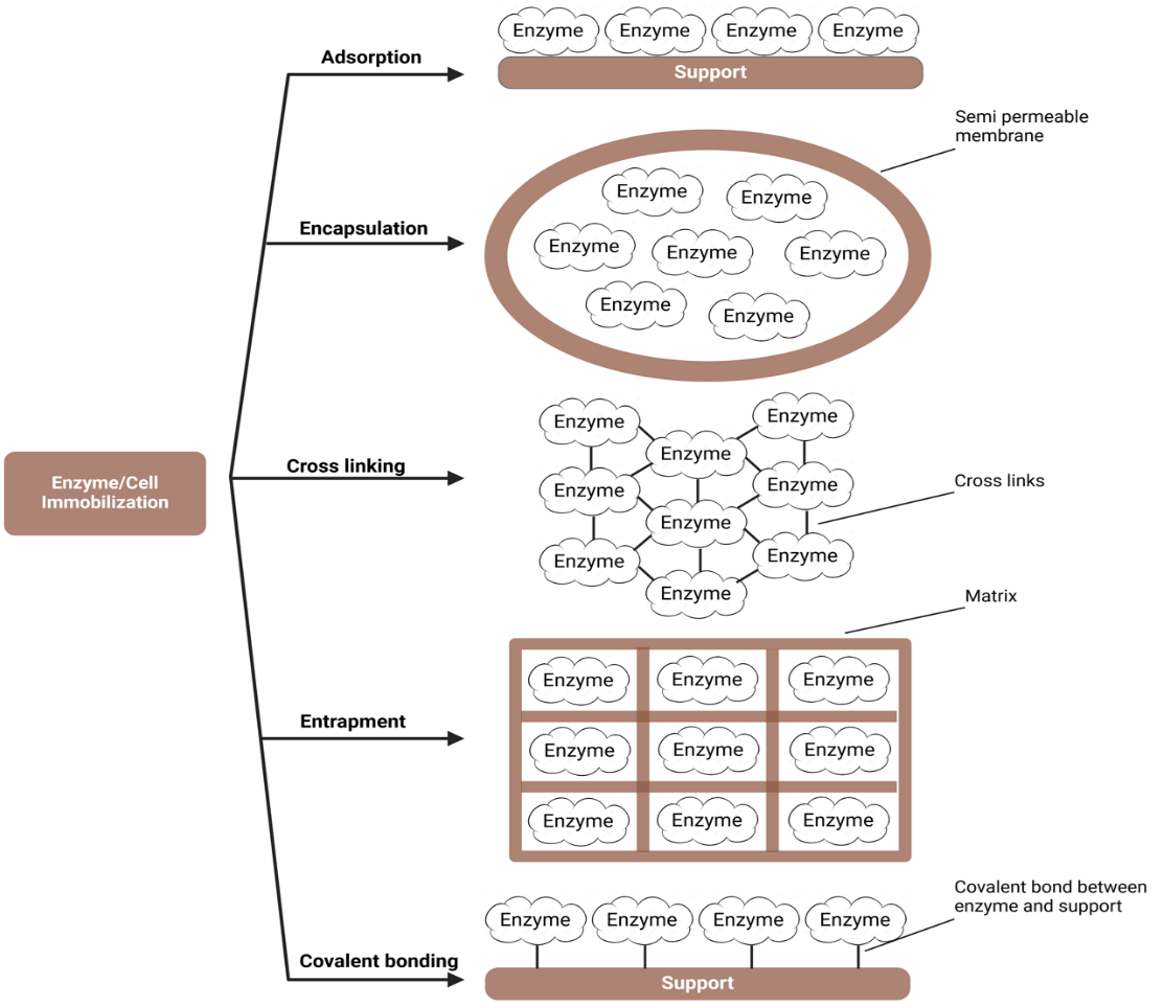
Figure 14.13. Scheme of various enzyme immobilization techniques. Mohammadi and Balan (CC0 1.0).
Table 14.3: Immobilized enzymes for food applications (16).
| Enzyme | Immobilization methods | Support | Application | Results |
|---|---|---|---|---|
| Protease | Entrapment | Mesoporous silica and zeolite | Milk coagulant for dairy products | Immobilized protease converted milk in cheeses in < 90 min |
| Protease | Cross-linking | Chitosan beads | Production of gluten-eliminated beer | Gluten content was decreased from 65 to 15 mg/kg |
| Amylase | Covalent bonding | Chitosan | Barely malt hydrolysis | Increasing the product yield |
| Pectinase | Encapsulation | Polyvinyl alcohol gel | Apple juice clarification | 80% turbidity reduction |
| Pectinase | Adsorption/cross-linking | Polymeric matrix | Apple juice clarification | Immobilized pectinase was recycled 10 times |
| Lipase | Encapsulation | α-lactalbumin nanotubes | Flavor enhancement for low-fat cheeses | Immobilization enhanced the flavor of cheese |
| Lipase | Adsorption | Hydroxyapatite | Production of methyl acetate aroma | Immobilized lipase activity was preserved after 3 h at 60 oC |
| β-glucosidase | Covalent binding | Chitosan/silica | Lactose hydrolysis in dairy products | High efficiency of lactose hydrolysis reaching up to 62% |
| β-glucosidase | Covalent binding | Polyvinyl alcohol | Lactose hydrolysis in dairy products | High efficiency of lactose hydrolysis reaching up to 75% |
| β-glucosidase | Encapsulation | Calcium alginate | Hydrolysis of cheese whey lactose | High efficiency of lactose hydrolysis reaching up to 72% |
14.7. Screening for Novel Enzymes
The range of biochemical reactions facilitated by enzymes is extensive. When developing an industrially relevant biocatalytic process, the first rate-limiting step is to identify suitable enzyme candidates. Conventionally, novel biocatalysts are obtained by cultivating microbial strains in pure form and subsequent screening examinations. However, evidence from molecular ecology shows that standard laboratory cultivation methods yield less than 1% of the diverse species found in certain complex niche microbiomes. while environments such as soil can harbor up to 10,000 unique organisms.
The significant discrepancy between the observed microbial abundance in nature and the number of viable colonies cultured under laboratory conditions, known as the ‘plate count anomaly’, has led to the proposal to isolate microbial DNA directly from the environment to obtain an unbiased genomic representation. This approach involves the direct cloning of the combined genomes of resident microflora, termed the metagenome, which provides an unfiltered snapshot of genomic content. Metagenomics has proven to be effective in the discovery of new biocatalysts from the uncultured environmental microbiota. Metagenomic libraries have been generated in various studies to discover new molecules or biocatalysts for various biotechnological purposes. Metagenomics has revealed a wealth of new genes, ranging from small genes encoding enzymes to complex gene clusters producing proteins, using different vector systems.
The strategies for obtaining biocatalysts by culturing or cloning metagenomes are shown schematically in Fig. 14.14 (17). In the case of cultivation (upper part), only part of the existing microbial diversity can be cultivated and grown in pure culture. Cultivable microorganisms can be characterized and subjected to fermentation processes, whereby the products are obtained directly or after isolation, cloning, and expression of the relevant genes. In contrast, non-cultivable microorganisms are only accessible via their extracted genomes. In this approach (lower part), the collective genomes of all microorganisms in each habitat (the metagenome) are extracted and cloned into surrogate host systems for subsequent screening and product expression (18).
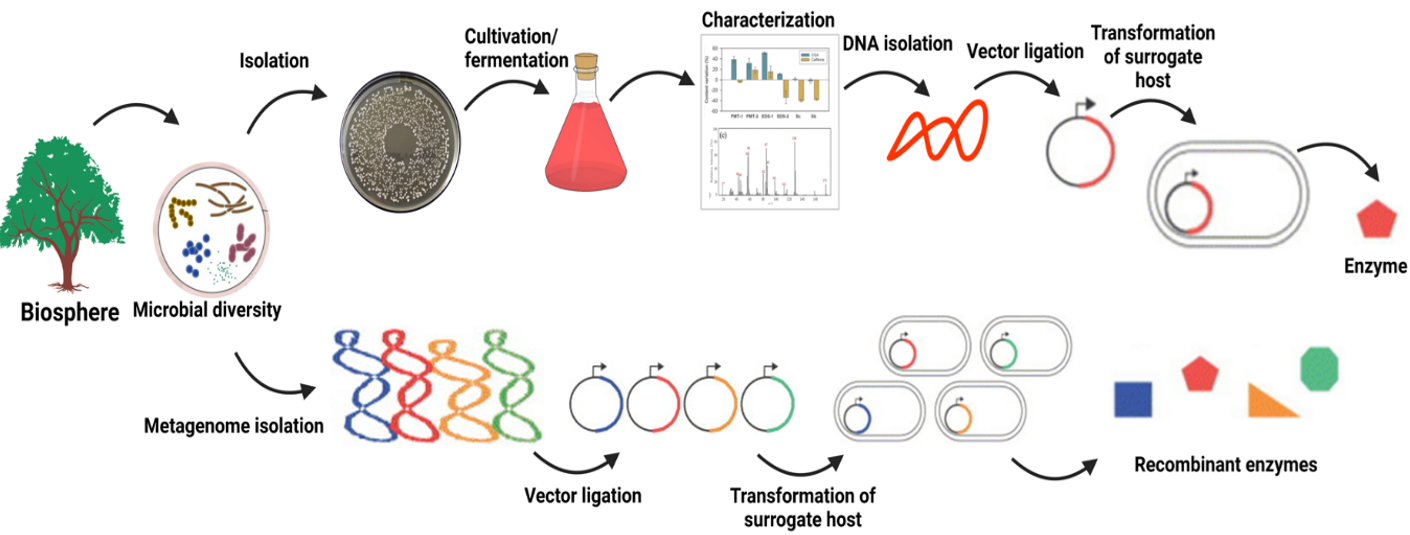
Figure 14.14. A simplified contrast between the cultivation approach, which involves isolating and screening individual microorganisms for enzymatic activity, and the metagenomic approach, which seeks out novel biocatalysts by extracting and studying microbial DNA directly from environmental samples. Mohammadi and Balan (CC0 1.0).
Applications of Industrial Enzymes
Enzymes with important industrial applications include carbohydrate-cleaving enzymes such as cellulases, amylases and invertases, proteolytic enzymes such as trypsin, chymotrypsin and papain as well as lipases, which can hydrolyze various lipids and fats. In addition, proteolytic enzymes from bacteria and fungi are also of great importance. These enzymes are widely used in various sectors such as the food and beverage industry as well as in textile, paper and detergent production. Proteases in particular play a central role in beverage production, meat and leather processing, cheese production, the formulation of detergents and in the bread and confectionery industry. Lipases are used to modify lipids and fats, produce organic acids, including fatty acids, formulate detergents and manufacture products like coconut butter. In addition, enzymes play a central role in the chemical industries and serve as reagents in organic synthesis to facilitate stereospecific reactions.
14.8. Enzymes in the Food Industry
Enzymes are innovative alternatives to conventional chemical or mechanical processes that improve yield, efficiency and quality in food processing. Their use in food production has a rich historical tradition dating back to ancient civilizations. During technological progress, a whole range of new types of enzymes have been discovered, which are used in various branches of industry and are constantly finding new applications. The extensive use of enzymes in food processing underlines their value, which is due to their specificity and catalytic abilities. The origins of the use of enzymes can be traced back some 8,000 years and can be seen in the production of staple foods such as beer, cheese, bread, wine and vinegar (19). Since then, the development and implementation of food processing techniques using enzymatic reactions has accelerated rapidly. Fig. 14.15 summarizes microbial enzymes with applications in the food industries. Some representatives of enzymes in the food industry are reviewed and discussed here (20).
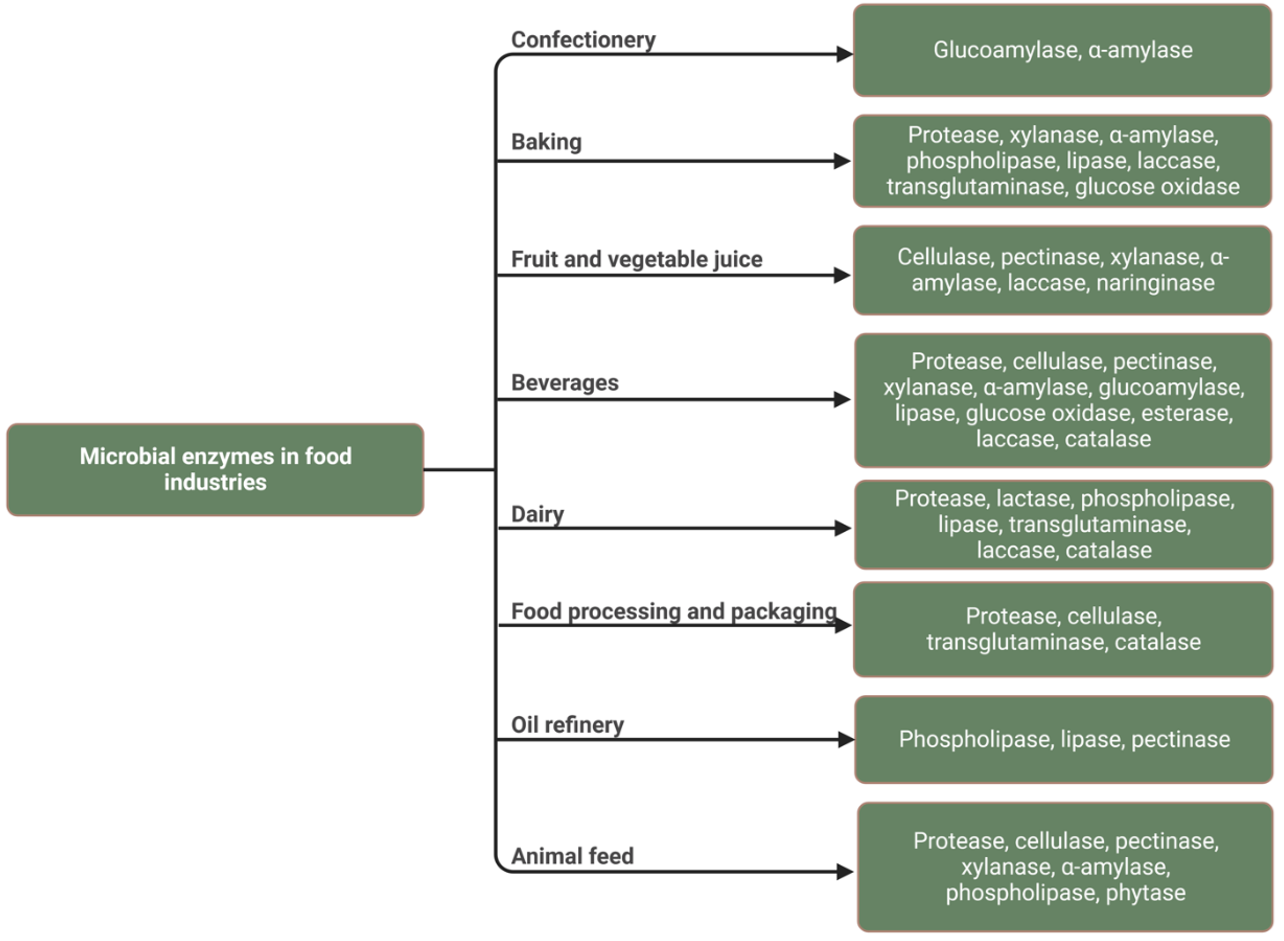
Figure 14.15. Microbial enzymes with applications in food industries. Mohammadi and Balan (CC0 1.0).
(i) Hydrolases: Hydrolases are an important group of enzymes that are widely used in food processing, as they enable hydrolysis reactions in which large macromolecules such as carbohydrates, proteins, lipids and polyphenols are broken down into smaller components through interaction with water. Several types of hydrolases play important roles in food manufacturing:
Alpha-amylases are a versatile enzyme for the food industry due to their ability to break down starch molecules by hydrolyzing alpha-1,4-glycosidic bonds to produce glucose, maltose and other short-chain dextrins. The diverse applications of α-amylases in the food industry include baking, brewing, starch liquefaction and as a digestive aid. In baking, the added alpha-amylase converts the starch in the dough into sugar and dextrin. Starch hydrolysis also improves dough conditioning and the volume of the finished bread. Alpha-amylases also act as anti-staling agent to improve the quality of the bread. In starch sweeteners, thermostable alpha-amylases from Bacillus species liquefy gelatinized corn and potato starch slurries by reducing viscosity and allowing further saccharification. This enzymatic conversion produces large-volume glucose and maltose syrups, which can be further fermented by specific microorganisms (e.g. Saccharomyces cerevisiae) to obtain the desired products, such as alcohol. In addition, specialty syrups and branched dextrin produced with the help of alpha-amylases serve as glazes, films and coatings in various cereal and confectionery products due to their solubility and moisture-binding properties. Alpha-amylases also help in the clarification of fruit juices by hydrolyzing starch, while working alongside pectinases and cellulases. This increases yield and production efficiency.
Lipases are enzymes that break down triglycerides into fatty acids and glycerol by hydrolysis. In the dairy industry, microbial lipases assist with cheese ripening by releasing specific flavor-imparting fatty acids during the ripening process. Lipases also hydrolyze triglycerides from milk fat to release free fatty acids that improve the flavor development of dairy products. Treating dough with lipase-modified milk fat can strengthen the dough structure and increase the volume of the loaf in baked goods. In addition, the use of lipases in combination with amylases can extend the shelf life of bread and reduce the glycemic response by interacting with lipids and starch during dough fermentation. Beyond baking, lipases are used in the production and enhancement of flavors in butter, margarine, ice cream, beer and wine. The ability of lipases to selectively hydrolyze ester bonds in triglycerides enables targeted flavor changes and the release of fatty acids in fatty foods and beverages through enzymatic reactions.
The enzyme lactase is another hydrolyzing enzyme which plays a key role in enabling people with lactose intolerance to consume dairy products by hydrolyzing lactose into glucose and galactose. Lactose intolerance is the inability to fully digest the milk sugar, lactose, due to low levels of the enzyme lactase in the small intestine. Manufacturers use lactase to produce low-lactose and lactose-free milk, yogurt and ice cream. In ice cream, lactase not only promotes digestibility, but also improves scoop ability and creaminess through partial lactose hydrolysis. Treatment with lactase can also increase the sweetness of dairy products, reducing the need for additional sweeteners. An important industrial use of lactase is the pretreatment of the lactose-rich cheese whey, a byproduct of cheese making, which enables profitable whey processing, as a rich protein source, and addresses environmental issues related to its disposal.
Proteases catalyze the hydrolysis of proteins by breaking peptide bonds. This makes them invaluable in the food industry when it comes to modifying the structure and functionality of proteins to improve the quality, taste, digestibility and processing efficiency of a variety of products. One of the major applications of protease in the food industry is in the process of cheese making where proteases, such as chymosin, are used to destabilize the casein micelles (milk protein), leading to milk coagulation to form the cheese curd. Other applications of proteases include accelerating cheese ripening by generating flavors from milk proteins, shortening dough mixing times and improving the texture of baked goods by cleaving gluten networks, tenderizing meat by hydrolyzing connective tissue, improving beer fermentation by optimizing amino acid balance even at low pH using acidic fungal proteases, and improving protein solubility and digestibility through controlled hydrolysis.
(ii) Oxidoreductases: Oxidoreductase comprises various enzymes such as peroxidase, reductase, dehydrogenase, oxidase, oxygenase and hydroxylase. These enzymes play a crucial role in the conversion of common sugars such as glucose and sucrose into valuable products. In addition, oxidoreductase demonstrates the ability to degrade aflatoxins, toxic metabolites produced by certain species of Aspergillus. This enzyme effectively degrades aflatoxins in pistachio nuts. Oxidoreductases are also used to preserve the freshness of seafood, improve baked goods and pasta, and extend the shelf life of foods through enzymes such as glucose oxidase, which is increasingly used in the baking industry for its dough-firming and oxidative effects. In various food applications, glucose oxidase improves taste, flavor, stability and shelf life by removing glucose and oxygen, especially in diabetic beverages and food packaging.
(iii) Isomerases: The complex process of producing rare sugars such as D-arabinose, L-fucose and L-ribulose, which are scarce and expensive to obtain, has attracted considerable attention in the food industry. Given the lucrative commercial prospects, researchers have been looking at various methods of sugar synthesis, with a clear focus on enzyme-driven approaches in recent years. Sugar processing typically involves the use of elevated temperatures to accelerate the reaction rate, minimize the risk of microbial contamination, improve the solubility of the sugar and achieve optimal product yield. To solve the problem of enzyme denaturation at high temperatures, scientists are exploring the potential of thermophilic enzymes derived from organisms adapted to extreme heat conditions.
Glucose isomerase, a key enzyme in food processing, facilitates the conversion of D-glucose to D-fructose, a process that is critical to produce high fructose corn syrup (HFCS). HFCS is known for its excellent solubility, sweetness and cost-effectiveness compared to sucrose and is widely used in the food and beverage industry. Isomaltulose is another promising sugar alternative characterized by properties such as slow digestion, sustained energy release, low carrier formation potential and a low glycemic index. In the large-scale production of isomaltulose, sucrose is enzymatically converted from beet sugar with the help of sucrose isomerase. In addition, L-arabinose isomerase has attracted attention due to its role in the synthesis of rare sugars. The enzymatic isomerization catalyzed by arabinose isomerase enables the production of rare sugars such as D-tagatose, L-ribulose and L-ribose, which are rarely found in nature.
(iv) Lyases: Lyases are a class of enzymes that catalyze reactions involving the breaking or formation of chemical bonds, resulting in the elimination of a group from the substrate. One notable application of lyases in the food industry is the modification of flavors and aromas. Terpene lyases, for example, are enzymes that can catalyze the cleavage of terpenes, aromatic compounds found in many plant foods. This enzymatic activity can be used to enhance or alter the sensory properties of foods, contributing to the development of unique and desirable flavors in beverages, baked goods and other consumer products.
Lyases are also used in the production of certain food additives. Pectate lyases, for example, are enzymes involved in the breakdown of pectin, a complex polysaccharide found in plant cell walls. Pectin is found in tomatoes, apple, pineapple, orange, lemon pulp, orange peel and other citrus fruits. Lyases break down pectin into smaller sugar units, reducing its gel-forming ability. The result is clearer, easier to extract juices and concentrates with higher yields. In fruit juice processing, pectate lyases are used to facilitate the extraction of juice by breaking down pectin, resulting in an improved juice yield. This enzymatic action not only aids the extraction process but also contributes to the clarity and stability of the finished fruit juices. Pectin liquors also help to produce smoother jams and jellies with the desired texture and prevent unwanted gelling. They can also clarify wines and beers by removing the pectin that causes cloudiness.
(v) Transferases: Transferases are a class of enzymes that facilitate the transfer of functional groups such as sugars, phosphates, and methyl groups from one molecule to another. They have notable applications in the food industry, with transglutaminase serving as a prominent example. Transglutaminase plays a crucial role in the production of protein components for a variety of foods. When incorporated into skim milk through microbial processes, transglutaminase increases the resilience of yogurt by partially binding casein molecules, thus increasing its breaking strength. Its application also extends to bakery products, where it promotes the cross-linking of gluten proteins and forms robust high molecular weight polymers that strengthen the bread structure, increase dough volume, and improve the elasticity of the bread crumb. In addition, in meat products, transglutaminase improves water retention, binding efficiency, consistency, and texture, softening the bland appearance of meat while maintaining its sensory properties. Another vital transferase is glucosyltransferase, which can transfer glucose molecules to various acceptors to increase sweetness, alter the properties of starch, and even produce prebiotics for gut health. This versatile enzyme plays a key role in producing syrups, sweeteners, and yogurts with just the right sweetness and texture. Transferases can even clarify beverages in which pectin methyl esterase modify pectin, reducing its tendency to gel and form cloudiness. They lead to clearer juices, wines, and beers and improve their visual appeal and storage stability.
14.9. Enzymes in the Biofuel Industry
Biofuels are categorized into different generations based on their feedstocks, with each generation addressing specific challenges and opportunities. First-generation (1G) biofuels, derived from starchy, sugary and oily food crops, such as sugarcane, corn, wheat, beets, and soybeans, are widely used. However, 1st generation biofuels compete with the food sector. In response to this limitation, second-generation (2G) biofuels have emerged, derived from forestry and agro-industrial wastes rich in cellulose, hemicellulose, and lignin, collectively referred to as lignocellulosic materials. Despite their potential, 2G biofuels are currently hampered by high processing costs, particularly in the pretreatment steps. In the subsequent generations, third generation (3G) biofuels, microalgae play an important role as feedstock to produce biodiesel.
In recent years, new technologies have been introduced to produce biofuels using enzymes as promising catalysts. Research and development efforts in this area aim to improve process efficiency, reduce the negative impact on the environment, and increase the overall quality of biofuel production. Utilizing a wide range of biomass sources with enzymes can promote cleaner industrial processes. The success of using enzymes with robust catalytic activity and stability in biofuel production is largely due to advances in enzyme development and protein engineering. Some of the widely used enzymes in the biofuel industry are discussed here.
The application of hydrolases is particularly significant in the conversion of biomass into biofuels, contributing to the development of sustainable and renewable energy sources. These enzymes specialize in breaking down complex molecules like carbohydrates and lipids into simpler units, paving the way for their conversion into biofuels. Some key types of hydrolases involved in biofuel production include amylases, cellulases, hemicellulases, and lipases.
Enzymes involved in the production of 1G biofuels
In the biofuel industry, amylases play a role in the conversion of starchy feedstocks and crops such as corn, cassava and sweet potatoes into fermentable sugars for sustainable first-generation biofuel production. Starch is a renewable carbohydrate polymer that serves as an energy storage molecule in plants and consists of polymer components- amylose and amylopectin. The complex polysaccharides amylose and amylopectin cannot be directly fermented to ethanol by industrial microbes. Amylases serve as the crucial biocatalysts that open these food reserves from starch to produce biofuels. α-Amylases (E.C. 3.2.1.1) first catalyzes the end hydrolysis of internal α-1,4-glycoside bonds, resulting in shorter oligosaccharides and linear maltodextrin chains during starch gelatinization. Glucoamylases (E.C. 3.2.1.3) then act on α-1,4- and α-1,6-bonds and release fermentable glucose. Together, these amylases efficiently and cost-effectively hydrolyze starch granules into high-purity fermentable sugars under mild conditions without salts or corrosive acids, which are the key advantages over chemical hydrolysis (Fig. 14.16a). The resulting simple hexose sugars, especially glucose, are easily converted into first-generation ethanol by fermenters such as Saccharomyces cerevisiae or Zymomonas mobilis using established industrial processes that have been optimized worldwide for decades (Fig. 14.16b).
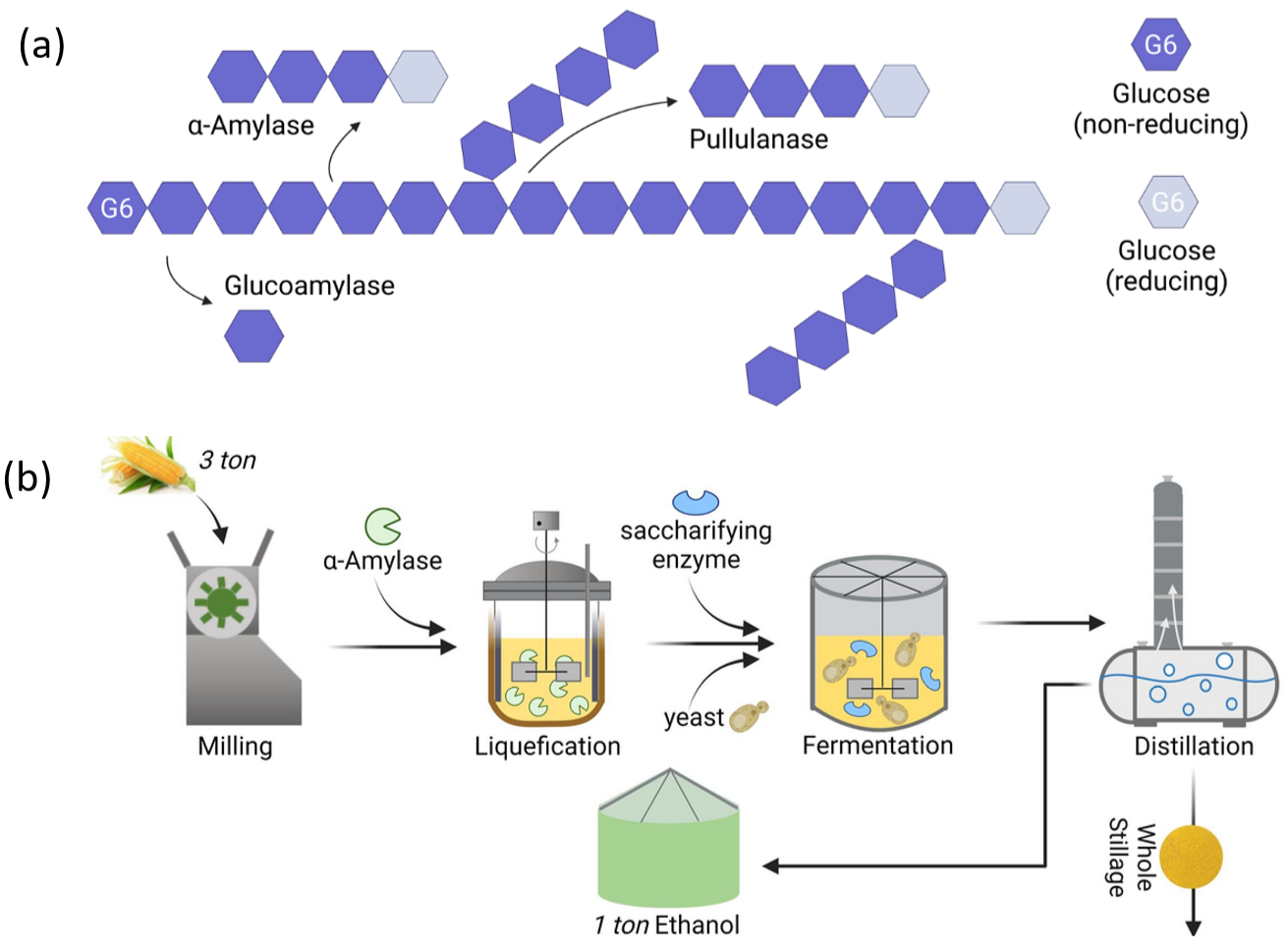
Figure 14.16. (a) Enzymes involved in biofuel production from starchy substrates and the reaction products. (b) Schematic representation of 1G bioethanol production process from corn, showing the main steps of milling, enzymatic liquefaction using α-amylase, saccharification, fermentation with yeast, and distillation. Reproduced with permission from Wang et al. (2023) (21) from Elsevier.
Process configurations for ethanol production include separate hydrolysis and fermentation (SHF), in which hydrolysis of lignocellulose to sugars and fermentation of sugars to ethanol take place in separate reactors, simultaneous saccharification and fermentation (SSF), in which enzymatic hydrolysis and fermentation of sugars are combined in a single reactor, which reduces capital costs, cold hydrolysis (CH), in which enzymatic hydrolysis is carried out at lower temperatures to reduce energy costs and eliminate the need for cooling before fermentation, and Consolidated BioProcessing (CBP), which aims to combine enzyme production, hydrolysis and fermentation in a single reactor using microorganisms that can both break down lignocellulose into sugar and ferment it into ethanol. This consolidates the entire process and eliminates the need for additional enzymes (Fig. 14. 17) (22).
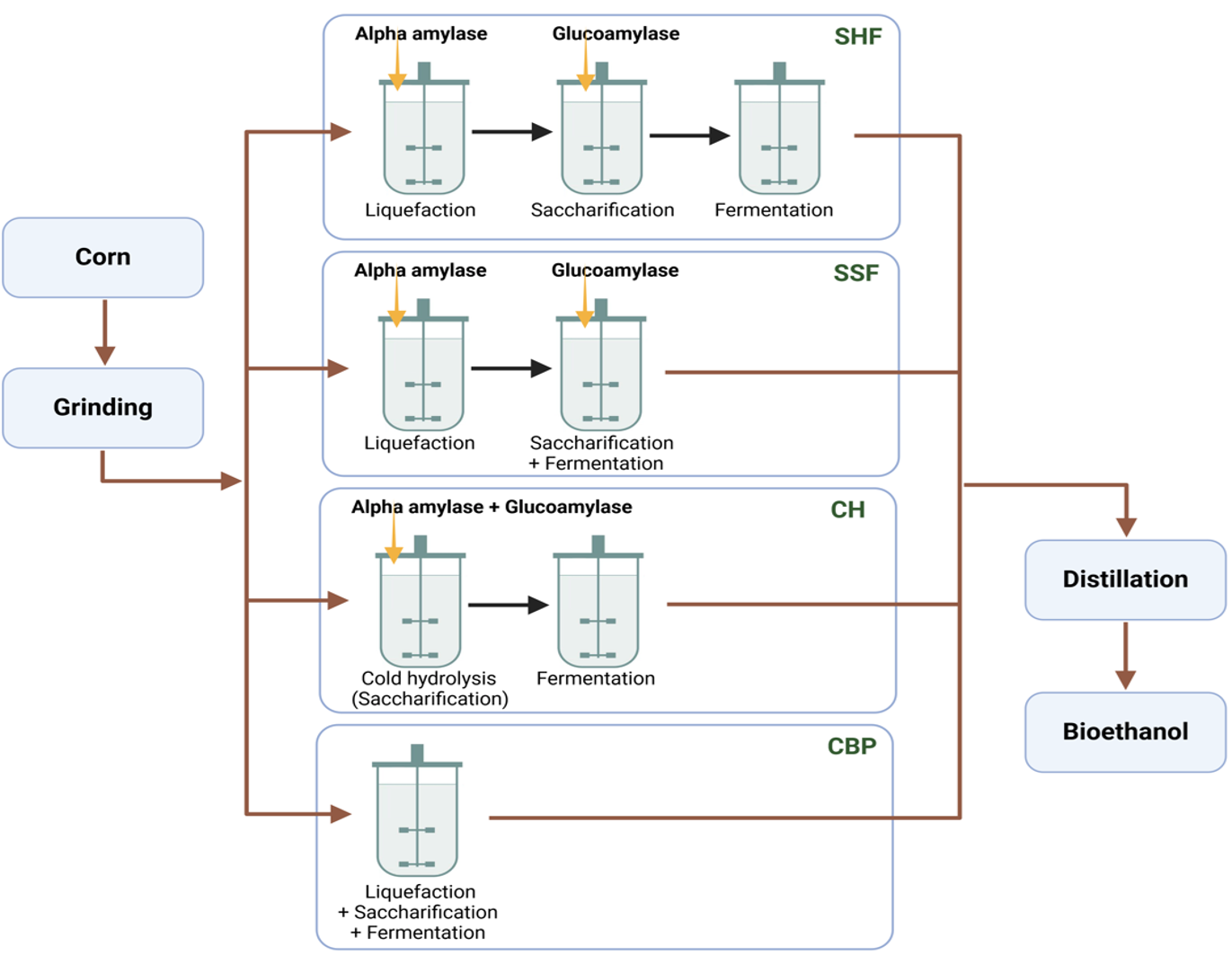
Figure 14.17. Process configurations for enzymatic hydrolysis and fermentation for ethanol production from starch. Mohammadi and Balan (CC0 1.0).
Some other common sources of 1G biofuels, such as sugarcane, sweet sorghum and sugar beet, contain large amounts of sucrose accumulated in their stems or tubers. This sucrose cannot be fermented directly into ethanol by industrial microbes. However, yeast and other ethanol-producing microorganisms can efficiently ferment hexose sugars such as glucose and fructose. Therefore, the sucrose in these feedstocks must be converted into fermentable simple sugars prior to bioconversion. This is where invertases are used, which catalyze the hydrolysis of sucrose into its constituent monosaccharides’ glucose and fructose. The fermentative microorganisms then metabolize these sugars to produce alcohols.
Lipases play an important role in the production of first-generation biodiesel from vegetable oils (e.g., soybean oil, palm oil, sunflower oil, olive oil, castor oil, etc.) and animal fats. Biodiesel is produced by transesterification of triacylglycerols (TAGs) from oils to generate fatty acid methyl esters (FAMEs), known as biodiesel. Lipases catalyze the transesterification reaction by breaking down the TAG molecules into FAMEs and glycerol. This conversion can be done via enzymatic transesterification using microbial lipases or chemical transesterification using alkaline catalysts. The lipases catalyze the critical first step of hydrolysis of triacylglycerols (TAGs) into free fatty acids (FFAs) and glycerol. The liberated FFAs can then be subjected to further esterification and transesterification reactions, often catalyzed by lipases themselves in non-aqueous systems, to synthesize FAME, the main components of biodiesel.
The versatility of lipases enables one-step (direct transesterification of TAGs) or two-step (TAG hydrolysis followed by FFA esterification) biodiesel production (Fig. 14.18) from a variety of lipid sources, including vegetable oil, animal tallow and cooking oils. However, due to food vs fuel debates, attention has been directed to the implementation of non-edible oils for biodiesel production which are discussed in the following sections.
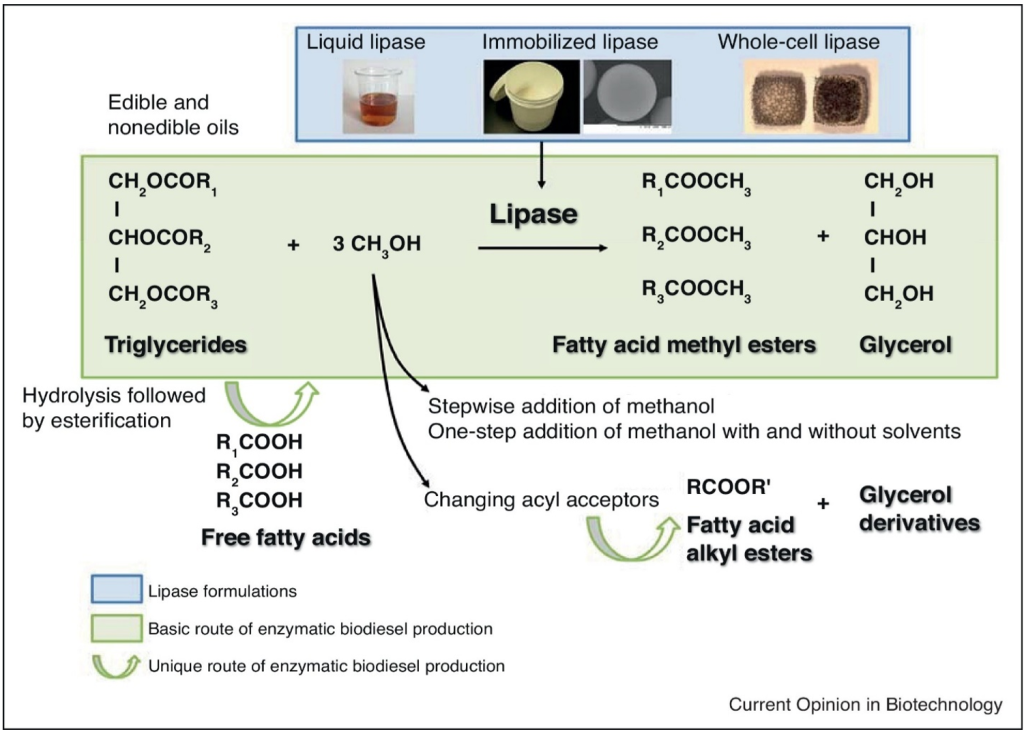
Figure 14.18. Overview of lipase-catalyzed biodiesel production pathways showing the transesterification of triglycerides to fatty acid methyl esters using different lipase formulations (liquid, immobilized, and whole-cell). Various process routes include one-step methanol addition and hydrolysis followed by esterification. Reproduced with permission from Hama et al. (2018) (23) from Elsevier.
Enzymes involved in the production of 2G biofuels
The complex and heterogeneous structure of lignocellulosic biomass, comprising of cellulose, hemicellulose and lignin, makes degradation to fermentable sugars extremely difficult (Fig. 14.19). No single enzyme can catalyze this degradation efficiently. Rather, a series of synergistic enzymes with complementary activities is required. These enzymes must work in tandem to hydrolyze cellulose, hemicellulose and lignin. By using cocktails, their combined action can access a wider range of bonds and sites in the plant cell wall architecture.
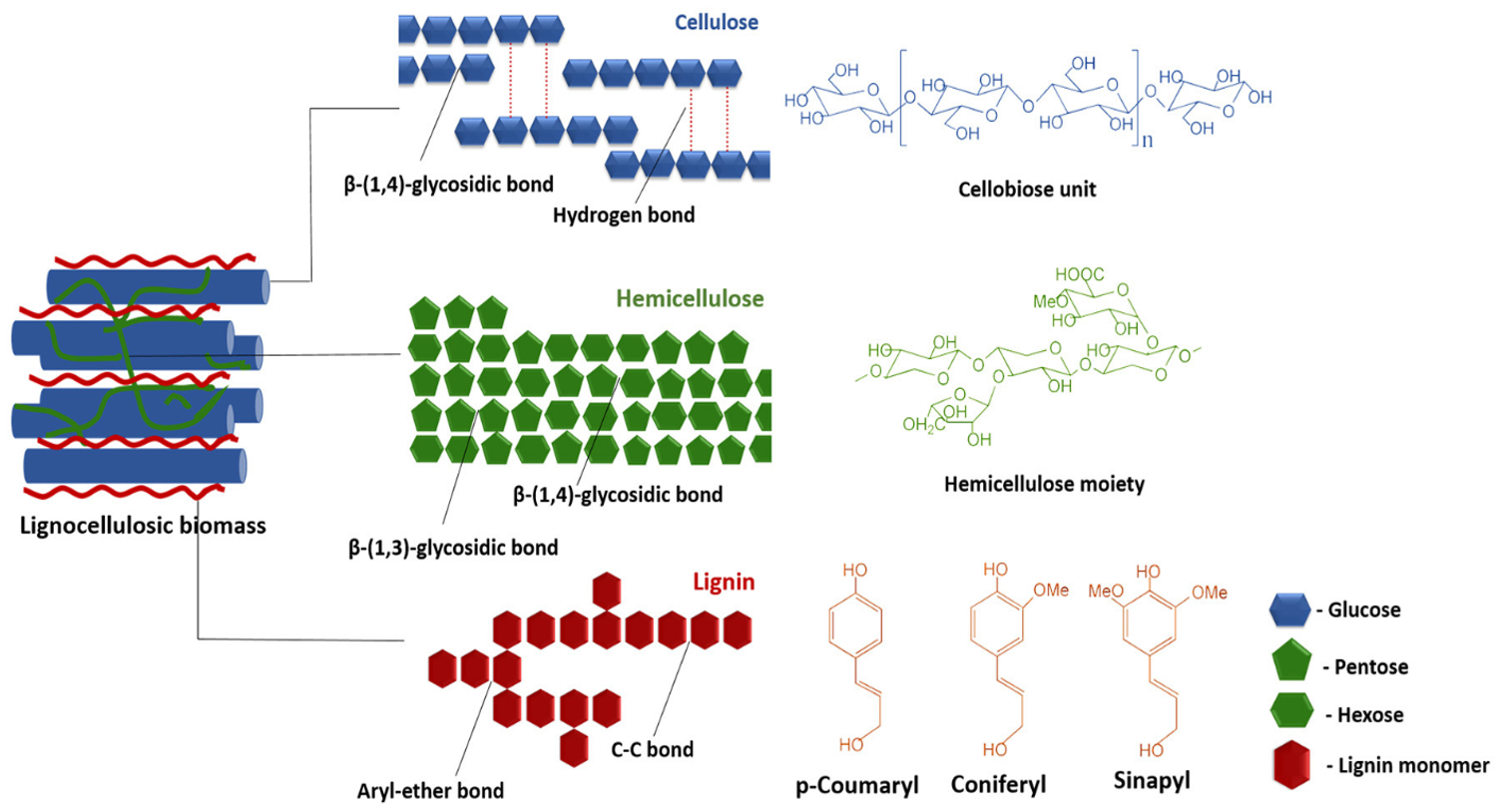
Figure 14.19. Molecular structure of lignocellulosic biomass showing the three main components. Here, cellulose (β-1,4-glycosidic bonds), hemicellulose (β-1,4-glycosidic and β-1,3-glycosidic bonds), and lignin (aryl-ether and C-C bonds) with their respective monomeric units are shown. Reproduced from Baruah et al. (2018) (24), under the Creative Commons Attribution License (CC BY).
Cellulases facilitate the crucial step of hydrolyzing recalcitrant cellulosic biomass into fermentable sugars for second-generation biofuels production. Cellulose is a linear polysaccharide consisting of β-1,4-linked D-glucose subunits. It is embedded in a complex lignocellulosic matrix that also contains hemicellulose and lignin in abundant and inexpensive agricultural residues such as sugar cane bagasse or corn stover. The rigid crystalline structure of cellulose makes it difficult for enzymes and microbes to break down. Cellulases act in a synergistic multienzymes system to provide access to cellulose conversion. Endoglucanases (E.C. 3.2.1.4) first attack internal amorphous sites and cleave them. Exoglucanases (E.C. 3.2.1.91) next attack the chain ends and release cellobiose or cello-oligosaccharides. Then β-glucosidases (E.C. 3.2.1.21) hydrolyze soluble cellodextrins and cellobiose to fermentable glucose (Fig. 14. 20a) (22). Together, these three cellulase classes efficiently break down the cellulosic fraction of the biomass into valuable C6 sugars for applications such as cellulosic ethanol production.
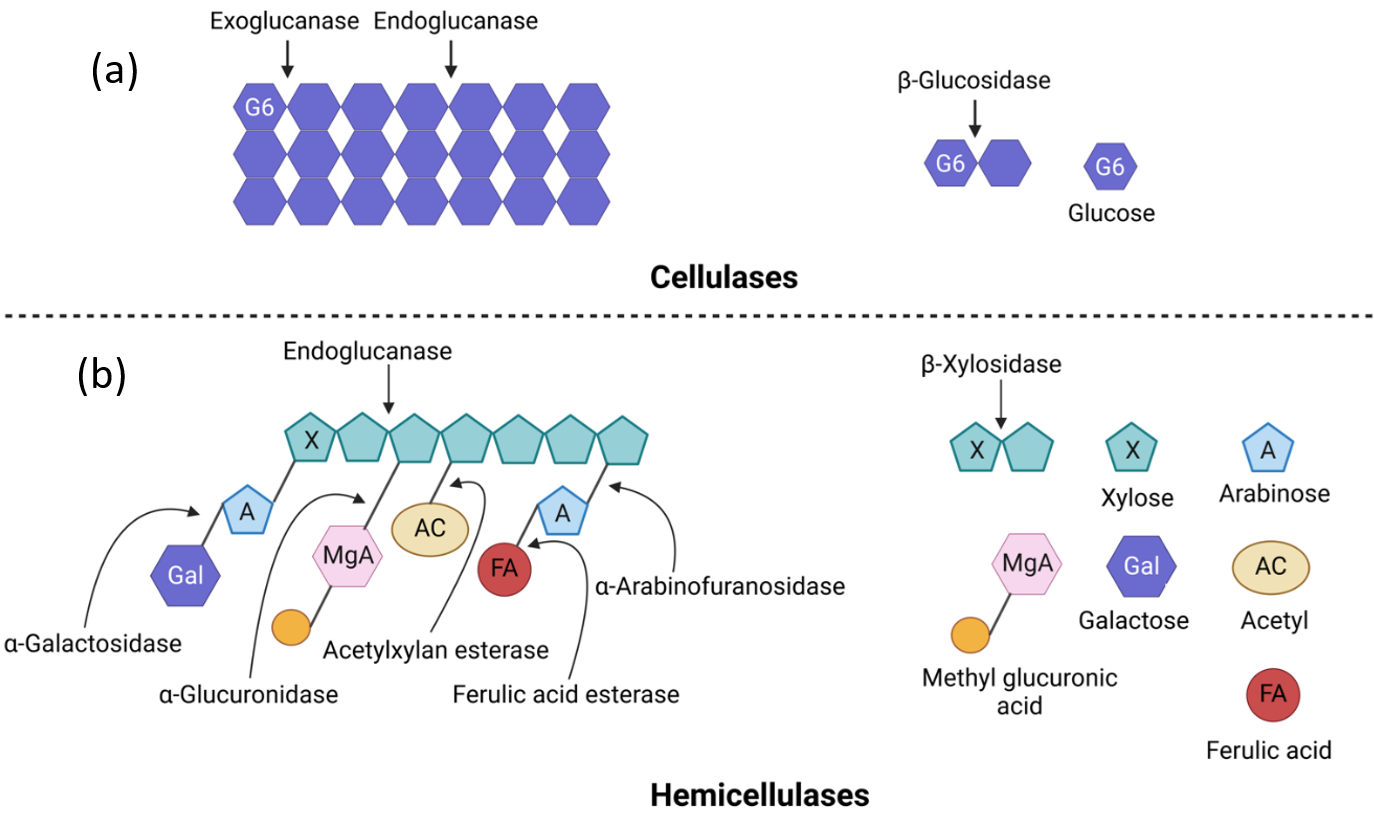
Figure 14.20. Enzymes involved in biofuel production from lignocellulosic substrates and the reaction products. Mohammadi and Balan (CC0 1.0).
Hemicellulases are a group of enzymes that break down hemicellulose, the second most common renewable polysaccharide after cellulose. The complete hydrolysis of hemicellulose is the key to unlocking the full sugar potential of plant biomass for biofuel production. Xylanases (endoxylanases, β-xylosidases) and accessory hemicellulases such as α-glucuronidases, acetyl xylan esterases and α-L-arabinofuranosidases work together to hydrolyze hemicellulose into its pentose and hexose sugar components (Fig. 14.20b). Hemicellulases also improve the efficiency of cellulases in the hydrolysis of biomass by improving the accessibility of cellulose. Although most industrial pretreatments already dissolve some of the xylan, the use of hemicellulases helps to release additional sugars, increasing the potential yields of biofuels from renewable plant biomass resources.
Lignin is a complex, aromatic and recalcitrant polymer that forms a protective barrier around the cellulose microfibrils. This coating provides mechanical strength and severely restricts cellulose polysaccharide digestion into fermentable sugars. Although lignin is highly resistant to chemical degradation, microbial ligninases such as laccase and peroxidases degrade the lignin and change its structure, making the biomass more accessible for saccharification. These ligninolytic enzymes use radical-based oxidative mechanisms to non-specifically break various ether and carbon-carbon bonds in the lignin polymer network. This makes the lignin more porous, soluble and fragmented, making the trapped cellulose more accessible. The use of ligninases minimizes the need for harsh acids, bases or heat pretreatment methods to overcome the repaicalcitrance of the biomass. The enzymatic hydrolysis of lignocellulosic biomass can be integrated with fermentation in various process configurations to optimize ethanol production. One common approach is the SHF process, where enzymatic saccharification and fermentation occur in separate vessels under their respective optimal conditions (Fig. 14.21).
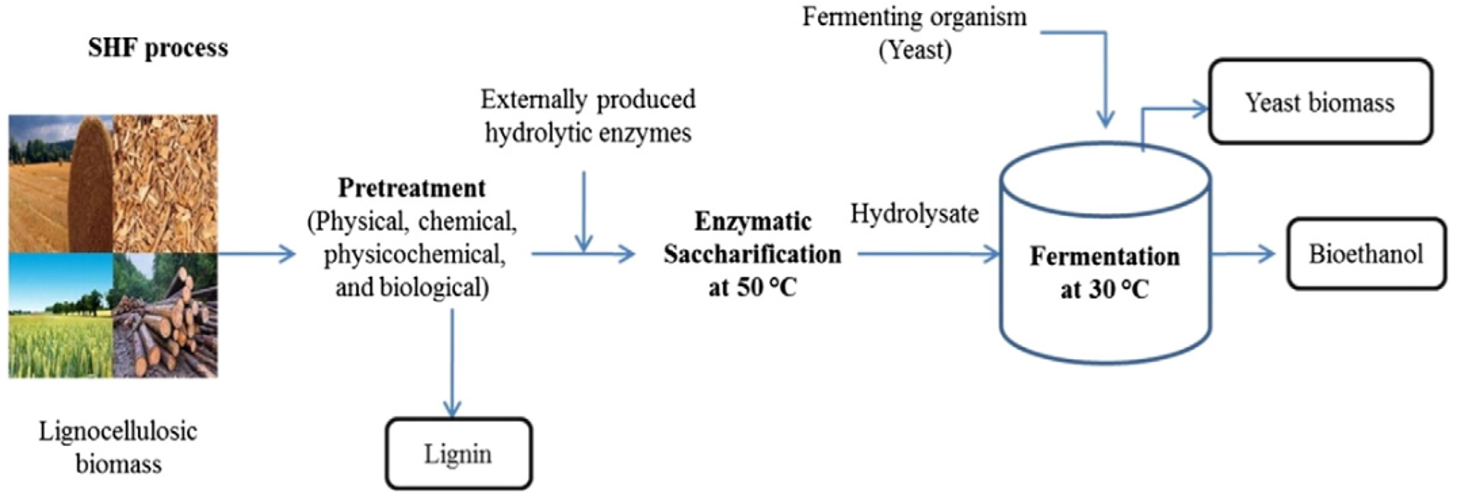
Figure 14.21. Schematic representation of the Separate Hydrolysis and Fermentation (SHF) process for bioethanol production from lignocellulosic biomass, showing sequential steps of pretreatment, enzymatic saccharification using externally produced hydrolytic enzymes, and fermentation with yeast. Reproduced from Choudhary et al. (2016) (25), distributed under the CC-BY license.
To efficiently produce second-generation bioethanol, a comprehensive bioconversion strategy is required that addresses each stage of processing. This includes careful selection of appropriate biomass feedstocks, application of effective pretreatment methods, optimization of enzymatic hydrolysis conditions, and implementation of efficient fermentation systems (Fig. 14.22). The cocktails of cellulases, hemicellulases and ligninases help to overcome the recalcitrance of lignocellulosic biomass in second-generation biofuel production. Optimizing these enzyme cocktails for different lignocellulosic feedstocks can maximize sugar yields for economical microbial fermentation to advanced biofuels such as cellulosic ethanol.
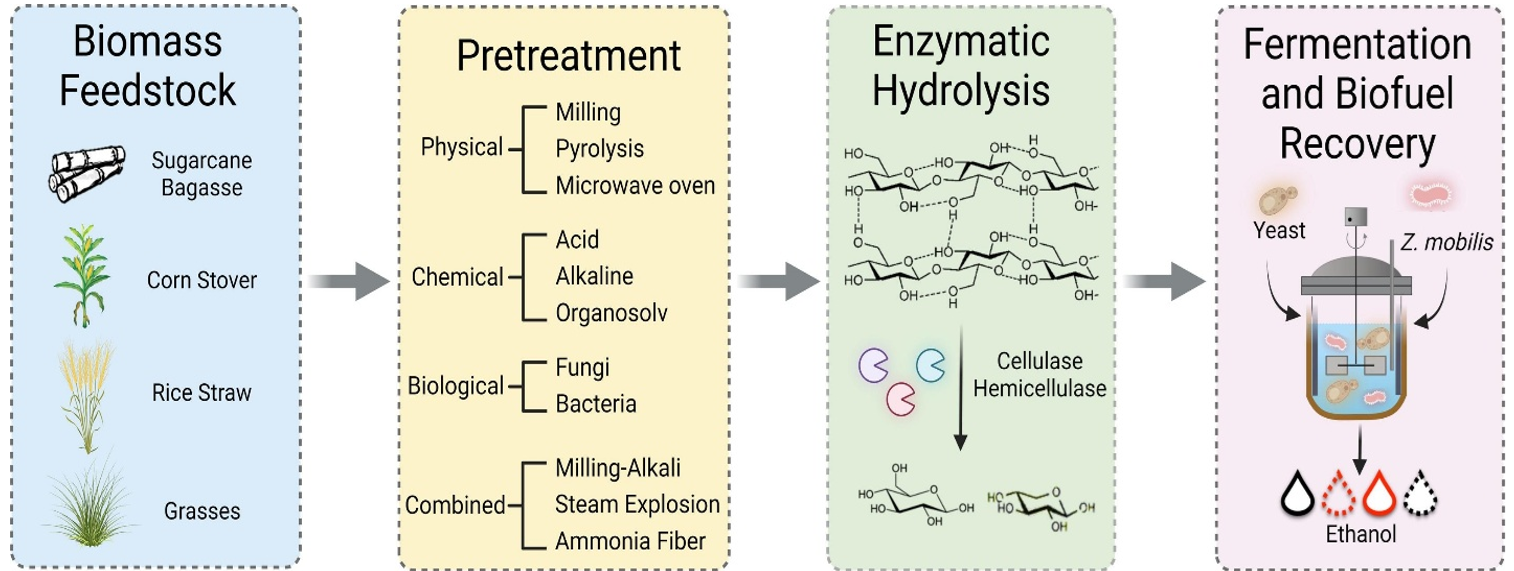
Figure 14.22. Schematic overview of second-generation bioethanol (cellulosic ethanol) production processes, highlighting the four major stages: biomass feedstock selection, pretreatment methods, enzymatic hydrolysis using cellulase and hemicellulase, and fermentation with yeast or Z. mobilis for ethanol recovery. Reproduced with permission from Wang et al. (2023) (21) from Elsevier.
Fig. 14.23 schematically illustrates the process configurations for enzymatic hydrolysis and fermentation for ethanol production from lignocellulosic biomass. The process configurations are like those explained for 1G biofuel production. An additional process is Simultaneous Saccharification and Co-Fermentation (SSCF) where both hexoses and pentoses can be converted in the same reactor (21).
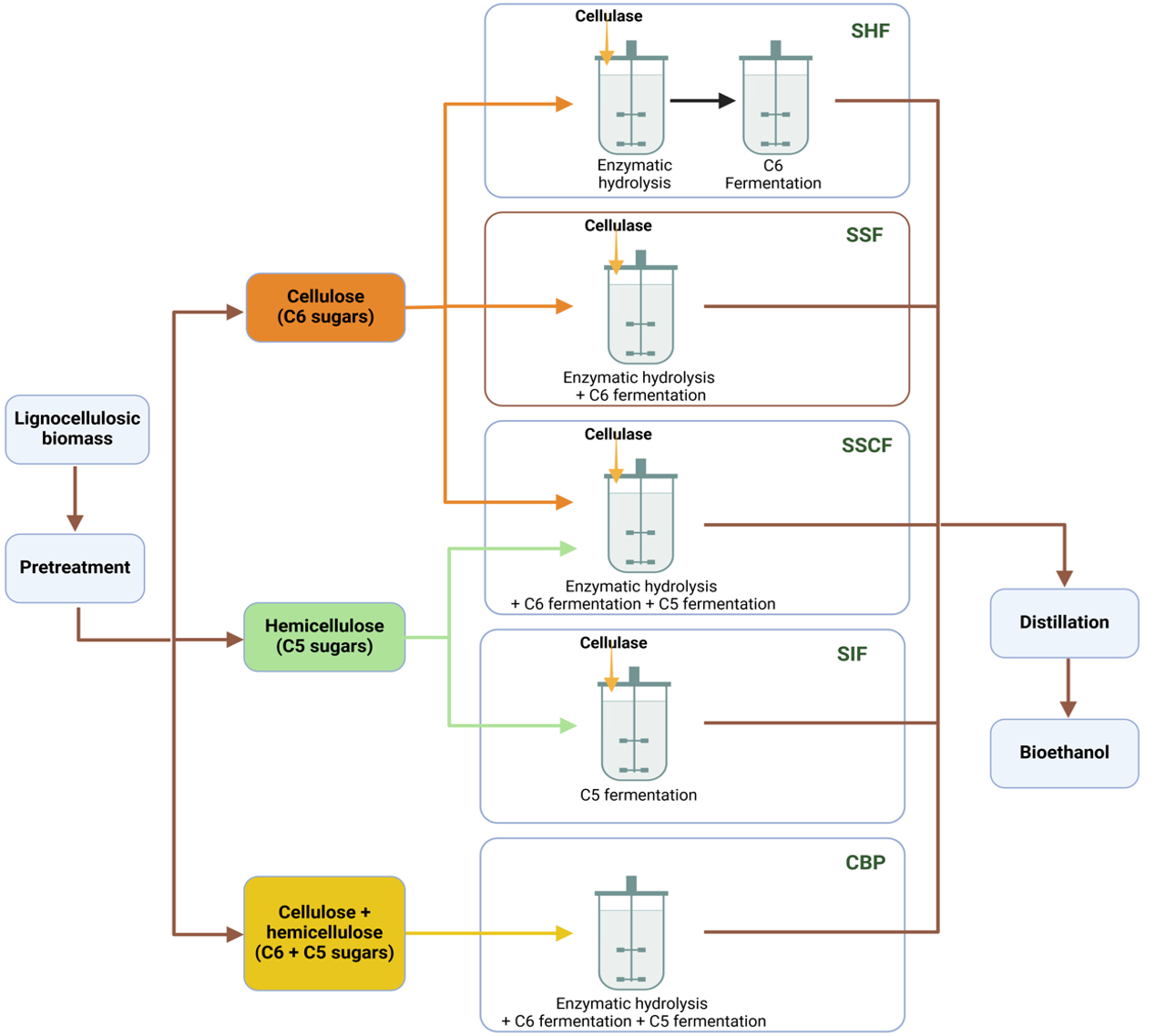
Figure 14.23. Process configurations for enzymatic hydrolysis and fermentation for ethanol production from lignocellulosic biomass. Mohammadi and Balan (CC0 1.0).
Enzymes involved in the production of 3G biofuels
Microalgae have attracted a lot of attention as potential feedstocks to produce third-generation biofuels (Fig. 14.24). These fast-growing photosynthetic microorganisms can accumulate high amounts of lipids, carbohydrates, and other valuable components. Unlike conventional crop-based feedstocks, microalgae don't compete with food production for arable land, can be cultivated in non-potable water (including wastewater and seawater), and exhibit significantly higher oil yields per hectare than terrestrial oilseed crops.
Figure 14.24. Overview of 3G biodiesel production process from microalgae, illustrating the complete cycle from cultivation using sunlight, CO₂, and nutrients from various wastewater sources, through extraction of algal oil, to biodiesel production. Reproduced with permission from Ali et al. (2024) (26) from Elsevier.
The conversion of microalgal biomass to biofuels involves several critical steps where enzymes play vital roles:
Figure 14.25. Five-step process of microalgae-based butanol production. Here, (1) cultivation of microalgae using sunlight and CO₂, (2) harvesting and drying of biomass, (3) enzymatic hydrolysis to release carbohydrates like sucrose, lactose, and maltose, (4) ABE (Acetone-Butanol-Ethanol) fermentation using Clostridium under anaerobic conditions, and (5) recovery of butanol as the final product. Reproduced with permission from Bauenova et al. (2024) (27) from Elsevier.
14.10 Enzymes in the Pharmaceutical Industry
Enzymes have emerged as powerful biocatalysts with diverse and valuable applications across the pharmaceutical industry (Fig. 14.26). These biological catalysts excel at catalyzing specific chemical reactions under mild conditions, while traditional chemical synthesis often requires harsh solvents, extreme temperatures, or toxic catalysts. Enzymes perform similar transformations in water, at room temperature, and with remarkable selectivity, providing greener alternatives to traditional chemical processes.
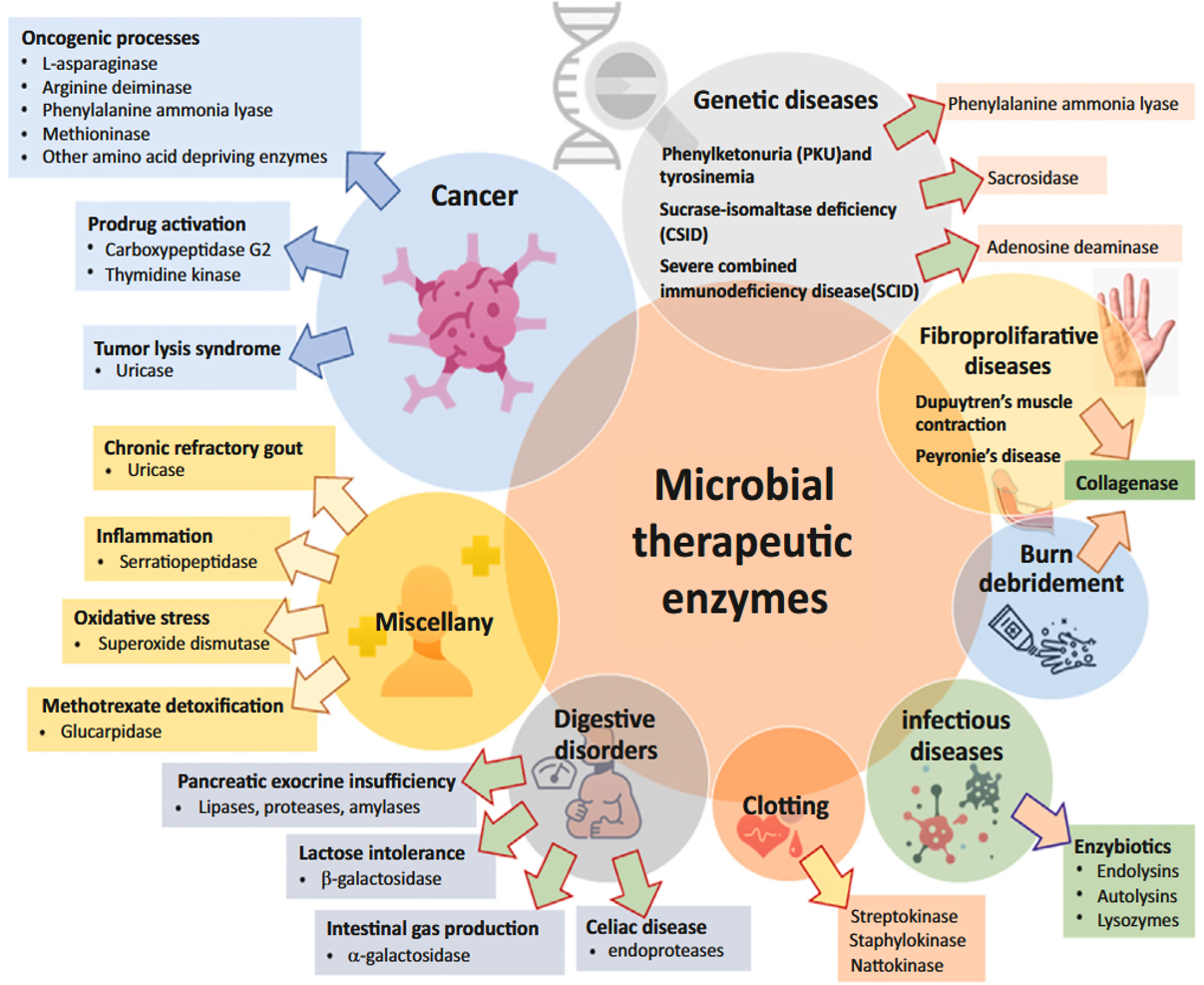
Figure 14.26. Therapeutic applications of microbial enzymes. Reproduced with permission from Arroyo et al. (2023) (28) from Elsevier.
The pharmaceutical industry has increasingly embraced enzymes as part of "green chemistry" initiatives, as enzymatic processes typically generate less waste, consume less energy, and reduce the need for toxic chemicals compared to traditional methods. For example, the enzymatic production of sitagliptin, a medication used to treat diabetes, reduced waste by 19% compared to the chemical approach, while simultaneously improving yield and product purity (29). Enzymes catalyze specific reactions with fewer side products, resulting in higher yields and less waste, while improving atom economy as enzyme-catalyzed reactions typically incorporate a higher percentage of starting materials into the final product. Since enzymes themselves are biodegradable proteins, they leave no harmful residues, and many enzymatic reactions can be performed in water rather than organic solvents. This document highlights key areas where enzymes contribute to pharmaceutical advances.
An important class of therapeutic enzymes is the "enzybiotics"; enzymes that can kill bacteria by breaking down their cell walls. These include:
- Lysozymes: Found naturally in tears and saliva, these enzymes can break apart bacterial cell walls and are used in some medical treatments for their antibacterial properties.
- Lysins: Derived from viruses that infect bacteria (bacteriophages), these enzymes precisely target specific bacterial species while leaving beneficial bacteria unharmed.
- Autolysins: These bacterial-derived enzymes that act on the producer bacterial cell and cleave the amide link between N-acetyl muramic acid and L-alanine in the peptidoglycan layer.
- Bacteriocins: Small antimicrobial peptides synthesized by both Gram-positive and Gram-negative bacteria that form networks in target cell membranes, disrupting their integrity.
- Unlike traditional antibiotics, bacteria rarely develop resistance to these enzymes because they target fundamental structural components of the bacterial cell wall (30).
Cancer treatment enzymes
Several enzymes have shown promise in cancer therapy (31):
- L-Asparaginase: Used to treat certain leukemias by depleting asparagine, an amino acid that cancer cells need but cannot produce themselves. It is particularly effective against acute lymphoblastic leukemia (ALL). L-ASNase rapidly stops the proliferation of cancerous cells that need high amounts of asparagine.
- L-Glutaminase: Shows inhibitory effects against various cancer cell lines including Vero, HeLa, MCF7, JURKAT, and HCT-116 RAW by targeting glutamine metabolism critical for cancer cell proliferation.
- L-Methioninase: Effective against various cancer types including breast, kidney, colon, lung, and prostate cancer by depleting methionine which many cancer cells are dependent on. Methionine dependency is described as an efficient biochemical phenomenon among cancerous cells not typically seen in normal cells.
- Lysine oxidase: Shows dose-dependent cytotoxic activity against cancer cell lines, particularly breast cancer MCF7, erythromyeloblastic leukemia K562, and colon cancer LS174T with documented IC50 values.
- Phenylalanine ammonia-lyase: Demonstrates effectiveness against breast and prostate cancer by targeting amino acid metabolism in cancer cells. It shows substantial decreases in cell viability during cytotoxicity experiments with these cancer types.
Other therapeutic enzymes
Additional therapeutic enzymes include (31):
- Clot-dissolving enzymes (like Activase): Used to treat heart attacks and strokes by breaking down blood clots that block arteries.
- Collagenase: Applied to wounds to help remove dead tissue and promote healing in burns and skin ulcers.
- Serratiopeptidase: Used for its anti-inflammatory, antibiofilm, analgesic, and antiedemic properties.
- Superoxide dismutase: Helps prevent precancerous cells, aging, ischemia, rheumatoid arthritis, and neurodegenerative diseases through its antioxidant and anti-inflammatory properties.
- Digestive enzymes: Supplements that help patients with pancreatic insufficiency properly digest their food.
- Uricase: Treats gout and hyperuricemia by breaking down uric acid.
Conclusions
Enzymes are widely used in the modern food and biofuel industries because of their ability to carry out chemical reactions with high specificity under relatively mild conditions. In the food industry, enzymes play a crucial role in the production of food and beverages such as bread, cheese, beer, wine, fruit, and vegetable products. The most important applications of enzymes include the processing of starch, the processing of proteins, the modification of fat and oil as well as extraction and clarification. Enzyme catalysis leads to improved yields, consistency, nutritional value, and cost savings in food production. New innovations in enzymes continue to create exciting opportunities for customized ingredients and food processing techniques. Similarly, enzymes have enabled more efficient, sustainable processes for biofuel production. Enzymes break down renewable feedstocks such as corn, sugarcane, oilseeds, cellulosic biomass, and microalgae into fermentable sugars that can be converted into bioethanol, biodiesel, and other biofuels. The use of enzymes enables higher yields and more environmentally friendly approaches compared to conventional chemical processes. In the future, enzymes will continue to serve as biocatalysts for the environmentally friendly and high-yield production of fuels and important food products. Advances in enzyme engineering, immobilization, and process integration will further exploit the potential of enzymes. Their specificity and efficiency under mild conditions perfectly position industrial enzymes as sustainable catalysts in various industries.
References
- H. Nevalainen, Chapter 7: Strategies and Challenges for the Development of Industrial Enzymes Using Fungal Cell Factories, Grand Challenges in Fungal Biotechnology, in: Powell’s Books, Springer, 2024.
- Rajiv. Dutta, Fundamentals of biochemical engineering, (2008) 292.
- N. A. J., Introduction to biotechnology and genetic engineering, Infinity Science Press LLC.
- K. Raval, T.G.-I.I.J. Basics, types and applications of molecular docking: A review, IP International Journal of Comprehensive and Advanced Pharmacology, 7 (2022) 12-16.
- Y. Valasatava, A. Rosato, N. Furnham, J.M. Thornton, C. Andreini, To what extent do structural changes in catalytic metal sites affect enzyme function?, Journal of Inorganic Biochemistry 179 (2018) 40–53. https://doi.org/10.1016/J.JINORGBIO.2017.14.002.
- Introduction to Chemistry: General, Organic, and Biological. https://2012books.lardbucket.org/books/introduction-to-chemistry-general-organic-and-biological/ (accessed February 11, 2024).
- S. Hornig, M. Michael, A. Grinyer, J. Turner, Introduction to enzymes and their applications, Introduction to Pharmaceutical Biotechnology, 2 (2018) 1-1-1–29. https://doi.org/10.1088/978-0-7503-1302-5CH1.
- D. Sutay Kocabaş, J. Lyne, Z. Ustunol, Hydrolytic enzymes in the dairy industry: Applications, market and future perspectives, Trends Food Science Technology 119 (2022) 467–475. https://doi.org/10.1016/J.TIFS.2021.12.013.
- A. Gessesse, F. Mulaa, S.L. Lyantagaye, L. Nyina-Wamwiza, B. Mattiasson, A. Pandey, Industrial Enzymes for Sustainable Bio-Economy: Large Scale Production and Application in Industry, Environment, and Agriculture in Eastern Africa, (2011). http://creativecommons. (accessed February 24, 2025).
- A. Gupta, P. Kumar, Microbial Oxidative Enzymes: Biotechnological Applications - Google Books, in: De Gruyter, 2024.
- S. Mukherjee, Isolation and Purification of Industrial Enzymes: Advances in Enzyme Technology, Advances in Enzyme Technology, First Edition (2019) 41–70. https://doi.org/10.1016/B978-0-444-64114-4.00002-9.
- A. Saxena, B.P. Tripathi, M. Kumar, V.K. Shahi, Membrane-based techniques for the separation and purification of proteins: An overview, Adv Colloid Interface Sci 145 (2009) 1–22. https://doi.org/10.1016/J.CIS.2008.07.004.
- K.S. Visan, R.J. Lobb, S. Ham, L.G. Lima, C. Palma, C.P.Z. Edna, L.Y. Wu, H. Gowda, K.K. Datta, G. Hartel, C. Salomon, A. Möller, Comparative analysis of tangential flow filtration and ultracentrifugation, both combined with subsequent size exclusion chromatography, for the isolation of small extracellular vesicles, Journal of Extracell Vesicles 11 (2022) 12266. https://doi.org/10.1002/JEV2.12266.
- S. Datta, L.R. Christena, Y.R.S. Rajaram, Enzyme immobilization: an overview on techniques and support materials, 3 Biotechnology 3 (2013) 1–9. https://doi.org/10.1007/S13205-012-0071-7/FIGURES/1.
- S. Adhikari, N. Pramanik, Application of Immobilized Enzymes in the Food Industry, Enzymes in Food Biotechnology: Production, Applications, and Future Prospects (2019) 711–721. https://doi.org/10.1016/B978-0-12-813280-7.00041-4.
- Y.R. Maghraby, R.M. El-Shabasy, A.H. Ibrahim, H.M.E.S. Azzazy, Enzyme Immobilization Technologies and Industrial Applications, ACS Omega 8 (2023) 5184–5196. https://doi.org/10.1021/ACSOMEGA.2C07560/ASSET/IMAGES/LARGE/AO2C07560_0003.JPEG.
- P. Lorenz, K. Liebeton, F. Niehaus, J. Eck, Screening for novel enzymes for biocatalytic processes: accessing the metagenome as a resource of novel functional sequence space, Current Opinion in Biotechnology 13 (2002) 572–577. https://doi.org/10.1016/S0958-1669(02)00345-2.
- J. Yun, S. Ryu, Screening for novel enzymes from metagenome and SIGEX, as a way to improve it, Microbial Cell Factories 4 (2005) 1–5. https://doi.org/10.1186/1475-2859-4-8/FIGURES/1.
- A.N.M. Ramli, P.K. Hong, N.H.A. Manas, N.I.W. Azelee, An overview of enzyme technology used in food industry, Value-Addition in Food Products and Processing Through Enzyme Technology (2022) 333–345. https://doi.org/10.1016/B978-0-323-89929-1.00011-1.
- M.O. Okpara, M.O. Okpara, Microbial Enzymes and Their Applications in Food Industry: A Mini-Review, Advanced Enzyme Research 10 (2022) 23–47. https://doi.org/10.4236/AER.2022.101002.
- M. Wang, J. Qiao, Y. Sheng, J. Wei, H. Cui, X. Li, G. Yue, Bioconversion of corn fiber to bioethanol: Status and perspectives, Waste Management 157 (2023) 256–268. https://doi.org/10.1016/J.WASMAN.2022.12.026.
- M.D.N. Ramos, T.S. Milessi, R.G. Candido, A.A. Mendes, A. Aguiar, Enzymatic catalysis as a tool in biofuels production in Brazil: Current status and perspectives, Energy for Sustainable Development 68 (2022) 103–119. https://doi.org/10.1016/J.ESD.2022.03.007.
- S. Hama, H. Noda, A. Kondo, How lipase technology contributes to evolution of biodiesel production using multiple feedstocks, Curr Opin Biotechnol 50 (2018) 57–64. https://doi.org/10.1016/J.COPBIO.2017.14.001.
- J. Baruah, B.K. Nath, R. Sharma, S. Kumar, R.C. Deka, D.C. Baruah, E. Kalita, Recent trends in the pretreatment of lignocellulosic biomass for value-added products, Frontiers Energy Research 6 (2018) 419973. https://doi.org/10.3389/FENRG.2018.00141/PDF.
- J. Choudhary, S. Singh, L. Nain, Thermotolerant fermenting yeasts for simultaneous saccharification fermentation of lignocellulosic biomass, Electronic Journal of Biotechnology 21 (2016) 82–92. https://doi.org/10.1016/J.EJBT.2016.02.007.
- S.S. Ali, S.G. Mastropetros, M. Schagerl, M. Sakarika, T. Elsamahy, M. El-Sheekh, J. Sun, M. Kornaros, Recent advances in wastewater microalgae-based biofuels production: A state-of-the-art review, Energy Reports 8 (2022) 13253–13280. https://doi.org/10.1016/J.EGYR.2022.09.143.
- M.O. Bauenova, A.K. Sadvakasova, B.D. Kossalbayev, G. Yilmaz, Z. Huang, J. Wang, H. Balouch, D.E. Zaletova, M.A. Lyaguta, H.F. Alharby, S.I. Allakhverdiev, Optimising microalgae-derived butanol yield, International Journal of Hydrogen Energy 49 (2024) 593–601. https://doi.org/10.1016/J.IJHYDENE.2023.14.065.
- M. Arroyo, I. de la Mata, C. Barreiro, J.L. García, J.L. Barredo, Application of microbial enzymes as drugs in human therapy and healthcare, Biotechnology of Microbial Enzymes: Production, Biocatalysis, and Industrial Applications, Second Edition (2023) 341–373. https://doi.org/10.1016/B978-0-443-19059-9.00002-5.
- G. Li, J. bo Wang, M.T. Reetz, Biocatalysts for the pharmaceutical industry created by structure-guided directed evolution of stereoselective enzymes, Bioorganic & Medicinal Chemistry 26 (2018) 1241–1251. https://doi.org/10.1016/J.BMC.2017.05.021.
- A. Shrivastava, N. Shrivastava, P.K. Singh, Enzymes in Pharmaceutical Industry, Enzymes in Food Biotechnology: Production, Applications, and Future Prospects (2019) 591–602. https://doi.org/10.1016/B978-0-12-813280-7.00034-7.
- N.Y. Patel, D.M. Baria, D.S. Pardhi, S.M. Yagnik, R.R. Panchal, K.N. Rajput, V.H. Raval, Microbial enzymes in pharmaceutical industry, Biotechnology of Microbial Enzymes: Production, Biocatalysis, and Industrial Applications, Second Edition (2023) 375–403. https://doi.org/10.1016/B978-0-443-19059-9.00025-6.
End-of-Chapter Questions

